��Ŀ����
��10�֣����ɶ�����4��Ԫ�أ������⣩��ɵ�����X 4.83g�ܽ���1Lˮ�С��ü�ζ�������Һ�ĵȷ�������V��10.0mL��������0.028mol/L NaOH��Һ23.8mL������һ��ʱ�䣬���´���Һ��ȡ�ȷ�������V��10.0mL�����ζ�����0.028mol/L NaOH��Һ47.5mL�����ڶ��εζ�֮�����õ���Һ��Ϊ���ȷݣ���һ����Һ�м���MgSO4��Һ�����ɳ���5.2mg����ڶ�����Һ�м���BaCl2��Һ��������������Ϊ53.5mg��
1��ȷ��X�Ļ�ѧʽ��
2����дX��ˮ��Ӧ�Ļ�ѧ����ʽ��
3����pH��V��X��V��NaOH��Ϊ���껭���ڶ����ȷ�������pH�ζ���ͼ��
1��NO2SO3F��5�֣�
NaOH��HNO3��NaNO3��H2O NaOH��HSO3F��Na2SO4��H2O��
NaOH��HF��NaF��H2O 2NaOH��H2SO4��Na2SO4��2H2O��
Mg2����2F����MgF2�� SO42����Ba2����BaSO4�� Ba2����2F����BaF2��
2��NO2SO3F��H2O��HNO3��HSO3F HSO3F��H2O��H2SO4��HF���������У�����1.5�֣�
3��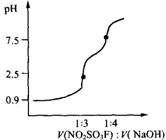 ��2�֣�
��2�֣�

��ϰ��ϵ�д�
�����Ŀ
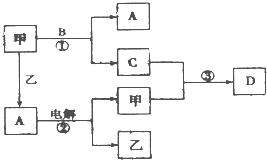 A��B��C��D���ɶ�����Ԫ����ɵ����ֳ����Ļ����DΪ����ɫ���壬�ס��������ֵ��ʣ���Щ���ʺͻ�����֮�������ͼ��ʾ��ת����ϵ��
A��B��C��D���ɶ�����Ԫ����ɵ����ֳ����Ļ����DΪ����ɫ���壬�ס��������ֵ��ʣ���Щ���ʺͻ�����֮�������ͼ��ʾ��ת����ϵ�� A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��ʾ����������²�ͬ����ش�
A��B��C��X����ѧ��ѧ�������ʣ����ɶ�����Ԫ����ɣ�ת����ϵ��ͼ��ʾ����������²�ͬ����ش�