��Ŀ����
CH4��H2��C�������ʵ���Դ���ʣ�����ȼ�յ��Ȼ�ѧ����ʽΪ��
��CH4(g)��2O2(g)=CO2(g)��2H2O(l)����H����890.3 kJ��mol��1��
��2H2(g)��O2(g)=2H2O(l)����H����571.6 kJ��mol��1��
��C(s)��O2(g)=CO2(g)����H����393.5 kJ��mol��1��
(1)����д���һ�ּ���ϸ������������øʹ������O2���ò���������������ϸ��ʹ1 mol��������CO2������Һ̬ˮ���ų�������________(���������������)890.3 kJ��
(2)������CO2�����ںϳɺϳ���(��Ҫ�ɷ���һ����̼������)��CH4��CO2=2CO��2H2��1 g CH4��ȫ��Ӧ���ͷ�15.46 kJ����������
����ͼ�ܱ�ʾ�÷�Ӧ�����������仯����________(����ĸ)��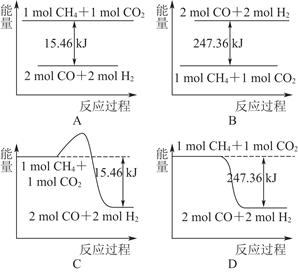
���������ʵ�����Ϊ1 mol��CH4��CO2����ij�����ܱ������У���ϵ�ų�����������ʱ��ı仯��ͼ��ʾ����CH4��ת����Ϊ________��
(3)C(s)��H2(g)����Ӧ������C(s)��2H2(g)=CH4(g)�ķ�Ӧ����ֱ�Ӳ�������ͨ��������Ӧ�������C(s)��2H2(g)=CH4(g)�ķ�Ӧ�Ȧ�H��________��
(4)Ŀǰ���������������ʵ��о���ȼ���о����ص㣬���й��������������ʵ��о������п��е���________(����ĸ)��
| A��Ѱ�����ʴ�����ʹCO2��H2O��Ӧ����CH4��O2�����ų����� |
| B��Ѱ�����ʴ������ڳ��³�ѹ��ʹCO2�ֽ�����̼��O2 |
| C��Ѱ�����ʴ���������̫����ʹ�����е�CO2�뺣���ɵ�CH4�ϳɺϳ���(CO��H2) |
| D������̬̼�ϳ�ΪC60����C60��Ϊȼ�� |
(1)����(2)��D����63%��(3)��74.8 kJ��mol��1��(4)C
����

 ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д��п�Ժ�����о����о�Ա���ʽ���������ͬ�к������Ա�������PM2.5��ѧ��ɼ���Դ�ļ��ڱ仯�о����֣�����PM2.5��6����Ҫ��Դ�����У�����β����ȼú�ֱ�ռ4%��18%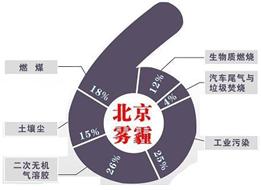
��1�����ھ�������β���ķ�ӦΪ��2NO(g)+2CO(g)
 2CO2(g)+N2(g)����֪�÷�Ӧ��570Kʱ��ƽ�ⳣ��Ϊ1��1059������Ӧ���ʼ���������˵����ȷ���ǣ�________
2CO2(g)+N2(g)����֪�÷�Ӧ��570Kʱ��ƽ�ⳣ��Ϊ1��1059������Ӧ���ʼ���������˵����ȷ���ǣ�________
| A��װ��β������װ�õ������ų��������в��ٺ���NO��CO |
| B�����β������Ч�ʵij��÷����������¶� |
| C������ѹǿ������ƽ�����ƣ���ʵ�ʲ����п�ͨ����ѹ�ķ�ʽ����侻��Ч�� |
| D�����β������Ч�ʵ����;����ʹ�ø�Ч���� |

 Ni(CO)4(g)������CO��Ӧ������������ж���Ϊ��ֹ�������ж�����ҵ�ϳ���SO2��ȥCO��������ΪS��CO2����֪��ط�Ӧ���̵������仯��ͼ��ʾ
Ni(CO)4(g)������CO��Ӧ������������ж���Ϊ��ֹ�������ж�����ҵ�ϳ���SO2��ȥCO��������ΪS��CO2����֪��ط�Ӧ���̵������仯��ͼ��ʾ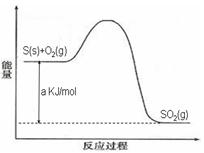
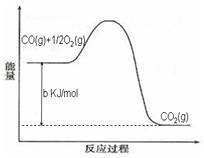
����SO2��ȥCO���Ȼ�ѧ����ʽΪ _____________________________________��
��3��NH3����ԭ�������SCR��������ĿǰӦ����㷺���������������ѳ������������Ļ�ѧ��Ӧ�ǣ�2NH3(g)+NO(g)+NO2(g)
 2N2(g)+3H2O(g)��H��0��Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�ǣ�������һ�֣�____________________��
2N2(g)+3H2O(g)��H��0��Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�ǣ�������һ�֣�____________________����4������ClO2�����������ﷴӦ�������£�

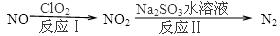
��Ӧ��Ļ�ѧ����ʽ��2NO+ClO2+H2O�TNO2+HNO3+2HCl����Ӧ������ӷ���ʽ�� ________________������11.2L N2���ɣ���״������������NO _________________ g��
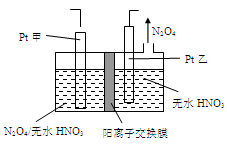
��5����ҵ�����к��е�NO2�����õ�ⷨ��������NO2Ϊԭ�Ͽ���������ɫ������N2O5���Ʊ�����֮һ���Ƚ�NO2ת��ΪN2O4��Ȼ����õ�ⷨ�Ʊ� N2O5��װ����ͼ��ʾ�� Pt��Ϊ _____��������������N2O5�ĵ缫��Ӧʽ��________________��
�й�������ŵ,��2020��,��λGDP������̼�ŷű�2005���½�40%��50%��
��1����Ч��̼���ֶ�֮һ�ǽ���,�������ⷽ������ܵ�������������
A.���ˮ����:2H2O 2H2��+O2��
2H2��+O2��
B.����ʹˮ�ֽ�����:2H2O 2H2��+O2��
2H2��+O2��
C.̫������ֽ�ˮ����:2H2O 2H2��+O2��
2H2��+O2��
D.��Ȼ������:CH4+H2O CO+3H2
CO+3H2
��2��CO2��ת�����л���ʵ��̼ѭ���������Ϊ1 L���ܱ�������,����1 mol CO2��3 mol H2,һ�������·�Ӧ:CO2��g��+3H2��g�� CH3OH��g��+H2O��g������H="-49.0" kJ/mol,���CO2��CH3OH��g����Ũ����ʱ��仯��ͼ��ʾ��
CH3OH��g��+H2O��g������H="-49.0" kJ/mol,���CO2��CH3OH��g����Ũ����ʱ��仯��ͼ��ʾ��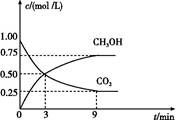
�ٴ�3 min��9 min,v��H2��=��������mol/��L��min����
����˵��������Ӧ�ﵽƽ��״̬�����������������ţ���
A.��Ӧ��CO2��CH3OH�����ʵ���Ũ��֮��Ϊ1��1����ͼ�н���㣩
B.���������ܶȲ���ʱ��ı仯���仯
C.��λʱ��������3 mol H2,ͬʱ����1 mol H2O
D.CO2����������ڻ�������б��ֲ���
��3����ҵ��,CH3OHҲ����CO��H2�ϳɡ��ο��ϳɷ�ӦCO��g��+2H2��g�� CH3OH��g����ƽ�ⳣ��:
CH3OH��g����ƽ�ⳣ��:
| �¶�/�� | 0 | 100 | 200 | 300 | 400 |
| ƽ�ⳣ�� | 667 | 13 | 1.9��1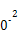 | 2.4��1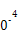 | 1��1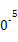 |
����˵����ȷ��������������
A.�÷�Ӧ����Ӧ�Ƿ��ȷ�Ӧ
B.�÷�Ӧ�������¶�ʱ,CH3OH��g�������������С,˵��v����CH3OH����С,v����CH3OH������
C.��T ��ʱ,1 L�ܱ�������,Ͷ��0.1 mol CO��0.2 mol H2,�ﵽƽ��ʱ,COת����Ϊ50%,���ʱ��ƽ�ⳣ��Ϊ100
D.��ҵ�ϲ����Ըߵ�ѹǿ��5 Mpa����250 ��,����Ϊ��������,ԭ����ת�������
(1)�״���һ����Ҫ�Ļ�����Ʒ�������ü״��������Ʊ���ȩ����ȩ����̬�״�ת����������ϵ��ͼ��ʾ��
��Ӧ�����е�������ϵ
�ټ״�������ת��Ϊ��ȩ�ķ�Ӧ��________(����ȡ����ȡ�)��Ӧ��
�ڹ��̢�����̢�ķ�Ӧ���Ƿ���ͬ��____________ԭ����__________________________________��
��д���״�������ת��Ϊ��ȩ���Ȼ�ѧ��Ӧ����ʽ________________________________��
(2)��֪����CH3OH(g)��H2O(g)=CO2(g)��3H2(g)����H����49.0 kJ��mol��1
��CH3OH(g)��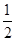 O2(g)=CO2(g)��2H2(g)����H����192.9 kJ��mol��1
O2(g)=CO2(g)��2H2(g)����H����192.9 kJ��mol��1
����˵����ȷ����________��
| A��CH3OHת���H2�Ĺ���һ��Ҫ�������� |
| B���ٷ�Ӧ�У���Ӧ�������������������������� |
C�����ݢ���֪��Ӧ��CH3OH(l)��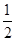 O2(g)=CO2(g)��2H2(g)�Ħ�H����192.9 kJ��mol��1 O2(g)=CO2(g)��2H2(g)�Ħ�H����192.9 kJ��mol��1 |
| D����Ӧ�ڵ������仯��ͼ��ʾ |


 CO2(g) + 3H2(g) ����H>0
CO2(g) + 3H2(g) ����H>0


 ,д��������Ӧ��Ӧ�Ļ�ѧ����ʽ:
,д��������Ӧ��Ӧ�Ļ�ѧ����ʽ:  CH3OH(g)
CH3OH(g)
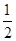 O2��g��=CO2��g������H2��b kJ��mol��1
O2��g��=CO2��g������H2��b kJ��mol��1 2NH3��g������H����92.4 kJ��mol��1����ʼ���ǽ�N2��H2�������20 mol�������1��1������5 L�ϳ����У���ӦǰѹǿΪp0����Ӧ������ѹǿ��p��ʾ����Ӧ������
2NH3��g������H����92.4 kJ��mol��1����ʼ���ǽ�N2��H2�������20 mol�������1��1������5 L�ϳ����У���ӦǰѹǿΪp0����Ӧ������ѹǿ��p��ʾ����Ӧ������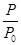 ��ʱ��t�Ĺ�ϵ��ͼ��ʾ��
��ʱ��t�Ĺ�ϵ��ͼ��ʾ��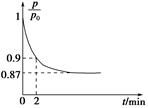
 2NH3(g) ��H����92.4 kJ��mol��1
2NH3(g) ��H����92.4 kJ��mol��1