��Ŀ����
��12�֣���һ�������£������Ϊ2 L�������м���2 mol O2��3 mol SO2���п��淴Ӧ��2SO2��g��+ O2��g��2SO3��g����2 min����O2�����ʵ���Ϊ1.6 mol����
��1��2 min�ڣ�SO2�����ʵ��������� mol��SO3�����ʵ��������� mol��
��2������O2��Ũ�ȱ仯����ʾ�÷�Ӧ�ķ�Ӧ���ʣ���V��O2���� ��
��3������SO3��Ũ�ȱ仯����ʾ�÷�Ӧ�ķ�Ӧ���ʣ���V��SO3���� ��
��1����6�֣�0.8 0.8 ��2����3�֣�0.1 mol/��L*min��
��3����3�֣�0.2 mol/��L*min��
����:

��ϰ��ϵ�д�
 Ӧ������ҵ��ϵ�д�
Ӧ������ҵ��ϵ�д�
�����Ŀ
 ��ҵ���Ʊ��ϳ����Ĺ�����Ҫ��ˮ�����������飺
��ҵ���Ʊ��ϳ����Ĺ�����Ҫ��ˮ�����������飺CH4 ��g��+H20��g���TC0��g��+3H2��g����H��0����һ�� �����£������Ϊ1L���ܱ������г���1mol CH4 �� g����1mol H20��g�������H2O��g����H2��g����Ũ����ʱ��仯 ��������ͼ��ʾ������˵����ȷ���ǣ�������
| A����ƽ��ʱ��CH4��g����ת����Ϊ75% | B��O-10 min �ڣ�v��CO��=0.075 mol?L-1��min-1 | C���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=O.1875 mol?L-1 | D����CH4��g��������������H20��g��������������ȣ���Ӧ����ƽ�� |
 2SO3��l����2min����O2�����ʵ���Ϊ1.6mol����
2SO3��l����2min����O2�����ʵ���Ϊ1.6mol����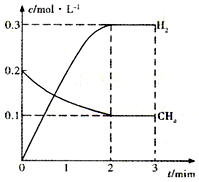 ��ҵ��������Ȼ������Ҫ�ɷ���CH4����H2O���и��������Ʊ��ϳ�����CO+H2����CO��H2��һ�����������Ʊ���ȩ�����ѵȶ����л����֪CH4��H2��CO��ȼ���ȣ���H���ֱ�Ϊ-890.3kJ?mol-1��-285.8kJ?mol-1��-283.0kJ?mol-1��18.0gˮ����Һ��ʱ�������仯Ϊ44.0kJ��
��ҵ��������Ȼ������Ҫ�ɷ���CH4����H2O���и��������Ʊ��ϳ�����CO+H2����CO��H2��һ�����������Ʊ���ȩ�����ѵȶ����л����֪CH4��H2��CO��ȼ���ȣ���H���ֱ�Ϊ-890.3kJ?mol-1��-285.8kJ?mol-1��-283.0kJ?mol-1��18.0gˮ����Һ��ʱ�������仯Ϊ44.0kJ��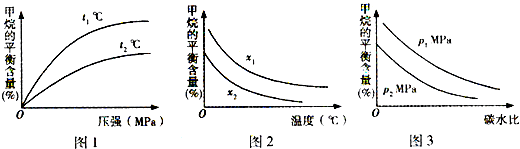
 2SO3��2minʱ�����O2�����ʵ���Ϊ1.6mol����
2SO3��2minʱ�����O2�����ʵ���Ϊ1.6mol����