��Ŀ����
��֪��
RCH2COOH��Br2 ![]() RCH��Br��COOH��HBr��R������
RCH��Br��COOH��HBr��R������
��һ���������Ϲ�ҵ�ķ����廯�������й���Ҷ������ϳ�·������ͼ��ʾ��
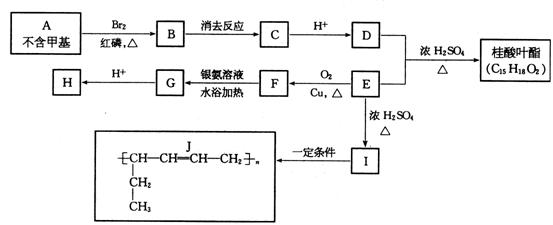
�Իش𣺣�1��I�Ľṹ��ʽ�� ��
E���Ӻ��еĹ������� ��д���ƣ���
��2��BgC�ķ�Ӧ������ �� FgG��Ӧ���� �����ţ���
a. ��ԭ��Ӧ b. ������Ӧ c. ȡ����Ӧ d. ��ȥ��Ӧ
��3��A�Ľṹ��ʽ�� ��
��4��д�����л�ѧ����ʽ��
EgF ��
D�����ļӾ۷�Ӧ ��
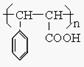
��5��д������Ҷ���Ľṹ��ʽ�� ��
��6��E��ͬ���칹��������ȩ���ҷ����к���3�����Ľṹ��_______�֡�
��1��![]() ��2�֣�
��2�֣�
�ǻ���̼̼˫�� ��2�֣�
��2��NaOH����Һ���� �� ��Ӧ���� b ������1�֣��ϼ�2�֣�
��3��A�Ľṹ��ʽ�ǣ�![]() �� ��2�֣�
�� ��2�֣�
��4����6�֣�
![]() + CuO ��
+ CuO �� ![]() +H2O+ Cu��
+H2O+ Cu��
D�ļӾ۷�Ӧ�� n ![]() ��
��
��5��![]() ��2�֣� (6) 3��2�֣�
��2�֣� (6) 3��2�֣�



 �����������ȡ��������ͬһ�������ϣ��ۿɷ���ˮ�ⷴӦ������һ��ˮ������ܷ���������Ӧ����һ��ˮ������������5�ֲ�ͬ��ѧ����������
�����������ȡ��������ͬһ�������ϣ��ۿɷ���ˮ�ⷴӦ������һ��ˮ������ܷ���������Ӧ����һ��ˮ������������5�ֲ�ͬ��ѧ����������
 ��д���Ա��Ӻ��Ҵ�Ϊԭ���Ʊ�
��д���Ա��Ӻ��Ҵ�Ϊԭ���Ʊ� �ĺϳ�·������ͼ
�ĺϳ�·������ͼ


 RCH��Br��COOH��HBr��R������
RCH��Br��COOH��HBr��R������


 F�Ļ�ѧ��Ӧ����ʽΪ
��
F�Ļ�ѧ��Ӧ����ʽΪ
��