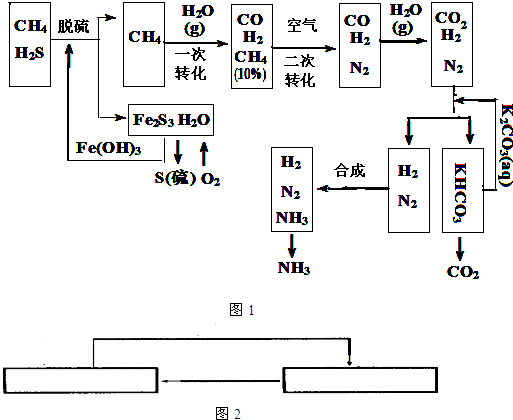
��Ŀ����
(10��)������Ȼ���ϳɰ��Ĺ�������ʾ�����£�

�����������̣����������գ�
��1����Ȼ������ʱ�Ļ�ѧ����ʽ��
��2��n mol CH4��һ��ת�������CO 0.9n mol������H2 mol���ú�n�Ĵ���ʽ��ʾ��
��3��K2CO3��aq���� CO2��Ӧ�ڼ�ѹ�½��У���ѹ������������ ����ѡ�۷֣�
��a����������ԭ�� ��b����ɳ����ԭ�� ��c������к�ԭ��
��4����KHCO3�ֽ�õ���CO2�������� ��д��CO2��һ����Ҫ��;����
��1��3H2S��2Fe(OH)3=Fe2S3��6H2O ��2��2.7n ��3��b
��4�������������������ȣ�����������Ҳ���֣�
��������
�����������1������ͼʾ��֪����Fe(OH)3��ȥ��Ȼ���е�H2S�����Է�Ӧ�Ļ�ѧ����ʽΪ3H2S��2Fe(OH)3=Fe2S3��6H2O��
��2��CH4+H2O=CO+3H2����������0.9n molCOʱ������H22.7nmol��
��3��K2CO3+H2O+CO2=2KHCO3����ѹƽ�������ƶ������ݵ�ԭ������ɳ����ԭ����
��4��CO2�����������ƴ����ԭ�ϡ�
���㣺��ҵ����
������������Ҫ����ѧ������������������ѶȽ�С���ۺ���ǿ��