��Ŀ����
�ƺͼ��ǻ��õļ����Ԫ�أ��ƺͼؼ��仯�������������������й㷺��Ӧ�ã���1����0.01mol�������ʢ�Na2O2��Na2O ��Na2CO3��NaCl�ֱ����100mL����ˮ�У��ָ������£�������Һ��������Ũ�ȵĴ�С˳���ǣ���Һ����仯���Բ��ƣ�
��2��NH3��NaClO��Ӧ�ɵõ��£�N2H4�����÷�Ӧ�Ļ�ѧ����ʽΪ
��3������һNa2CO3��NaHCO3�Ļ������Ʒ��ȡag�û�����ּ��ȣ�����bg����û������Ʒ��Na2CO3������������
��4���������ƣ�NaN3����ײ����ȫ�ֽ�����ƺ͵������ʿ�Ӧ����������ȫ���ң�������40.32L����״���£�������������Ҫ��������
��5����-�غϽ���ں˷�Ӧ���������Ƚ���Һ��5.05g��-�غϽ�����200mLˮ����0.075mol�����������Һ�����������ӵ����ʵ���Ũ����
��2������������ԭ��Ӧ�����غ�����жϲ������ƽ��ѧ����ʽ��ȼ�ϵ�أ�ȼ���ڸ���ʧ���ӷ���������Ӧ��
��3��NaHCO3���ȶ��������ֽ⣬���������ı仯���ò�ֵ�����
��4����Ӧ�ķ���ʽΪ2NaN3
| ||
��5����Ϸ�Ӧ�ķ���ʽ2Na+2H2O�T2NaOH+H2����2K+2H2O�T2KOH+H2�����㣮
�ʴ�Ϊ����=�ڣ��ۣ��ܣ�
��2��NH3��NaClO��Ӧ�ɵõ��£�N2H4������Ԫ�ػ��ϼ����߱���������������������������ԭΪ�Ȼ��ƣ���ѧ����ʽΪ2NH3+NaClO=N2H4+NaCl+H2O����--����ȼ�ϵ����һ�ּ��Ե�أ��õ�طŵ�ʱ����������ȼ�����ɵ�����ˮ�������ĵ缫��ӦʽΪ��N2H4-4e-+4OH-=N2+4H2O��
�ʴ�Ϊ��2NH3+NaClO=N2H4+NaCl+H2O��N2H4+4OH--4e-=4H2O+N2��
��3��NaHCO3���ȶ��������ֽ⣬����2NaHCO3
| ||
��2NaHCO3
| ||
168g 106g 62g
x bg
x=
| 168b |
| 62 |
����Ʒ��Na2CO3������Ϊag-
| 168b |
| 62 |
| 62a-168b |
| 62 |
���Ըû������Ʒ��Na2CO3������������
| 62a-168b |
| 62a |
�ʴ�Ϊ��
| 62a-168b |
| 62a |
��4����Ӧ�ķ���ʽΪ2NaN3
| ||
2NaN3
| ||
130g 67.2L
m 40.32L
m=
| 130g��40.32L |
| 67.2L |
�ʴ�Ϊ��78��
��5�����ơ��ص����ʵ����ֱ�Ϊa��b����Ӧ�ķ���ʽΪ2Na+2H2O�T2NaOH+H2����2K+2H2O�T2KOH+H2����
��a+b=0.075mol��2=0.15mol��
����Һ�����������ӵ����ʵ���Ũ����
| 0.15mol |
| 0.2L |
��23a+39b=5.05�������غ㣩��
��֮��a=0.050mol��b=0.10mol��n��Na����n��K��=1��2������-�غϽ�ѧʽΪNaK2��
�ʴ�Ϊ��0.75��mol/L����NaK2��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�������м��㣺
��1���������ƣ�NaN3����ײ���ֽ�����ƺ͵������ʿ�Ӧ����������ȫ���ң���78�˵���������ȫ�ֽ⣬������״���µ���
��2����-�غϽ���ں˷�Ӧ���������Ƚ���Һ��5.05g��-�غϽ�����200mLˮ����0.075mol������������Һ���������Ƶ����ʵ���Ũ��
��3������������Һ�����������ˣ��õ��������Ƶ���Һ�������Һ��ͨ�������̼�������з�Ӧ��2NaAl��OH��4+CO2��2Al��OH��3��+Na2CO3+H2O
��֪ͨ�������̼112L����״���£������ɵ�Al��OH��3��Na2CO3�����ʵ���֮��Ϊ4��5���������Һ��ͨ��Ķ�����̼Ϊ224L����״���£����������ɵ� Al��OH��3��Na2CO3�����ʵ��������ֵ��
��4��Ϊ�ⶨij�������պ�������������������е�Ԫ�ص������������ֽ���ͬ��������ι���ֱ���뵽50.00mL��ͬŨ�ȵ�����������Һ�У���ˮԡ����������ȫ���ݳ������¶�����β��ֽ⣩�������徭�������Ũ����������ȫ���ⶨŨ�������ӵ����������ֲⶨ������±���
| ������/g | 10.00 | 20.00 | 30.00 | 50.00 |
| Ũ�������ӵ�����/g | m | m | 1.29 | 0 |
����εijɷ�Ϊ
������е�Ԫ�ص���������Ϊ��
���ǻ��õļ����Ԫ�أ��Ƽ��仯�������������������й㷺��Ӧ�á�
������м��㣺
57����������(NaN3)��ײ����ȫ�ֽ�����ƺ͵������ʿ�Ӧ����������ȫ���ҡ�������40.32 L(��״����)������������Ҫ��������___g��
58����-�غϽ���ں˷�Ӧ���������Ƚ���Һ��5.05 g��-�غϽ�����200 mLˮ����0.075 mol������
(1)������Һ�����������ӵ����ʵ���Ũ��(������Һ����仯)��
(2)���㲢ȷ������-�غϽ�Ļ�ѧʽ��
59������������Һ�����������ˣ��õ��������Ƶ���Һ�������Һ��ͨ�������̼�������з�Ӧ��
2NaAl(OH)4+CO2��2Al(OH)3�� +Na2CO3+H2O
��֪ͨ�������̼336 L(��״����)������24 mol Al(OH)3��15 mol Na2CO3����ͨ����Һ�Ķ�����̼Ϊ112L����״����)���������ɵ�Al(OH)3��Na2CO3�����ʵ���֮�ȡ�
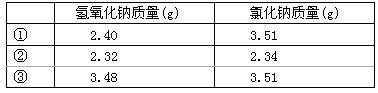
60�������£���ȡ��ͬ����������Ʒ����ˮ���������к���pH=7��Ȼ����Һ���ɵ��Ȼ��ƾ��壬���ɹ����в�Ʒ����ʧ��
| �� | ������������(g) | �Ȼ�������(g) |
| �� | 2.40 | 3.51 |
| �� | 2.32 | 2.34 |
| �� | 3.48 | 3.51 |
����ʵ��٢ڢ������������ƾ��������ʣ���ʵ�����ݿɿ���ͨ�����㣬�����ͱȽ��ϱ�3�����ݣ��������ۡ�
���ǻ��õļ����Ԫ�أ��Ƽ��仯�������������������й㷺��Ӧ�á�
������м��㣺
��1���������ƣ�NaN3����ײ���ֽ�����ƺ͵������ʿ�Ӧ����������ȫ���ҡ���78�˵���������ȫ�ֽ⣬������״���µ���___________________L ��
��2����-�غϽ���ں˷�Ӧ���������Ƚ���Һ��5.05 g��-�غϽ�����200 mLˮ����0.075 mol������������Һ���������Ƶ����ʵ���Ũ��______________________������Һ������仯����
��3������������Һ�����������ˣ��õ��������Ƶ���Һ�������Һ��ͨ�������̼�������з�Ӧ�� 2NaAl(OH)4+CO2��2Al(OH)3��+Na2CO3+H2O����֪ͨ�������̼112 L����״���£������ɵ�Al(OH)3��Na2CO3�����ʵ���֮��Ϊ4:5���������Һ��ͨ��Ķ�����̼Ϊ224L����״���£����������ɵ� Al(OH)3��Na2CO3�����ʵ��������ֵ��
��4��Ϊ�ⶨij�������պ�������������������е�Ԫ�ص������������ֽ���ͬ��������ι���ֱ���뵽50.00mL��ͬŨ�ȵ�����������Һ�У���ˮԡ����������ȫ���ݳ�(���¶�����β��ֽ�)�������徭�������Ũ����������ȫ���ⶨŨ�������ӵ����������ֲⶨ������±���
|
������/g |
10.00 |
20.00 |
30.00 |
50.00 |
|
Ũ�������ӵ�����/g |
m |
m |
1.29 |
0 |
�Իش�
����εijɷ�Ϊ_______________________________��
������е�Ԫ�ص���������Ϊ��_______________________________(����ʽ���㣩��