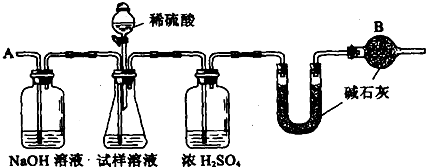
��Ŀ����
��֪ij���������к���NaCl���ʣ�Ϊ�ⶨ�����д��������������������ͼ�е�װ�ý���ʵ�顣����ʾ����ʯ������ʯ�����������ƵĻ�����������ˮ�Ͷ�����̼��

��Ҫʵ�鲽�����£��� ��ͼ��װ������������װ�õ�������
�� ��10.0 g����������ƿ�У�����������ˮ�ܽ⣬�õ�������Һ
�� ����ʢ�м�ʯ�ҵ�U�ܵ��������õ�20.0g
�� �ӷ�Һ©������6mol��L��1�����ᣬֱ�����ٲ�������ʱΪֹ
�� �ӵ���A����������һ�����Ŀ���
�� �ٴγ���ʢ�м�ʯ�ҵ�U�ܵ��������õ�22.0g
�� �ظ�����ݺ͢IJ�����ֱ��U�ܵ������������䣬Ϊ22.2g
����պͻش����⣺
��1��װ���и����B������ _________________________�����û�����Ӹø���ܣ����ԵĽ�� (��ƫ�ߡ�ƫ�ͻ䣩��
��2���������Һ©���е����ỻ��Ũ����ͬ�����ᣬ���ԵĽ�� (��ƫ�ߡ�
ƫ�ͻ䣩��
��3������ݵ�Ŀ���� ___________________�����û�н��в���ݵIJ��������ԵĽ��________________����ƫ�ߡ�ƫ�ͻ䣩��
��4�������д������������Ϊ___________________��
��1����ֹ�����е�CO2 ��H2O����U���У�2�֣���ƫ�ߣ�2�֣�
��2��ƫ�ߣ�2�֣�
��3���ѷ�Ӧ�е�CO2ȫ������U���У�2�֣���ƫ�ͣ�2�֣�
��4��53.0%
��������
�����������1��U���еļ�ʯ����Ϊ�����շ�Ӧ���ɵĶ�����̼����������Ҳ���ڶ�����̼�������B�����þ��Ƿ�ֹ�����еĶ�����̼��ˮ�ֽ���U�ܣ��Խ�������������е�ˮ�Ͷ�����̼����װ�ã�������Ϊ�Ƕ�����̼�����Զ�����̼������ƫ�������̼���Ƶ�����Ҳ��ƫ��ģ����Խ����ƫ��
��Ϊ����ֹ�����е�CO2��ˮ������U���У�ƫ�ߡ�
��2������������лӷ��ԣ�Ҳ�����Ŷ�����̼����U�ܣ�������Ϊ�Ƕ�����̼�����Զ�����̼������ƫ�������̼���Ƶ�����Ҳ��ƫ��ģ����Խ����ƫ��
��Ϊ��ƫ�ߣ�
��3�����ڷ�Ӧ������ƿ�д����ж�����̼������һ�����Ŀ������ǽ������Ķ�����̼��ȫ����U�ܣ�û�в���ݣ��в�����CO2û�з�Ӧ�����Խ��ƫ�͡�
��Ϊ���ѷ�Ӧ������CO2ȫ������U���У�ƫ�͡�
��4������Ҫ̼���Ƶ�����ΪX��
Na2CO3+H2SO4�TNa2SO4+H2O+CO2��
106 44
X 22.2-20.0
�б���ʽ�� ��
��
���X=5.3g��
���������д������������Ϊ
 ��100%=53.0%��
��100%=53.0%��
�𰸣�53.0%
���㣺̽�����ʵ���ɻ�������ʵĺ���������ʵ�鷽�������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�