��Ŀ����
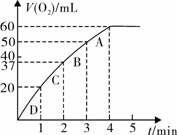
����0.10 mol MnO2��ĩ��50 mL H2O2��Һ�У��ڱ�״���·ų�����������ʱ��Ĺ�ϵ��ͼ��ʾ��

��д��H2O2�ڶ������������·�����Ӧ�Ļ�ѧ����ʽ ��
��ʵ��ʱ�ų������������� mL��
�۷ų�1/3��������ʱ��Ϊ min��
�ܷ�Ӧ�ų�3/4��������ʱ��ԼΪ min��
��A��B��C��D���㷴Ӧ���ʿ�����˳��Ϊ_____��____��____��____��
���ͷ�Ӧ���ʱ仯��ԭ�� ��
����H2O2�ij�ʼ���ʵ���Ũ��_____________�����뱣����λ��Ч���֣�
��2H2O2![]() 2H2O + O2������2�֣�
2H2O + O2������2�֣�
��60 mL �� ��1 min �� ����1�֣�
��2.5 min����1�֣� ��D>C>B>A����2�֣�
�����ŷ�Ӧ�Ľ��У�Ũ�ȼ�С����Ӧ���ʼ��� ��2�֣�
��0.11 mol��L��1����2�֣�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ