��Ŀ����
20����֪���и����Ȼ�ѧ����ʽ��C��s��+O2��g���TCO2��g����H1C��s��+$\frac{1}{2}$O2��g���TCO��g����H2
��S��s��+O2��g���TSO2��g����H1 S��g��+O2��g���TSO2��g����H2
��2H2��g��+O2��g���T2H2O��l����H1 2H2��g��+O2��g���T2H2O��g����H2
��CaCO3��s���TCaO��s��+CO2��g����H1 CaO��s��+H2O��l���TCa��OH��2��s����H2
���С�H1����H2 ���ǣ�������
| A�� | �٢� | B�� | �ڢ� | C�� | �٢� | D�� | �ڢ� |
���� ��̼������ȫȼ�շ��ȶ��ڲ���ȫȼ�շŵ��ȣ�
�ڹ������Ϊ��̬����Ҫ����������
��Һ̬ˮ��Ϊ��̬ˮ�Ĺ��������ȹ��̣�
��CaCO3��s���TCaO��s��+CO2��g����Ӧ���ȡ�H1��0��CaO��s��+H2O��l���TCa��OH��2��s�����ȡ�H2��0��
��� �⣺��̼������ȫȼ�����ɶ�����̼���ȶ��ڲ���ȫȼ������һ����̼�ŵ��ȣ���Ӧ���ʱ��Ǹ�ֵ���ʡ�H1����H2���ʴ���
�����ʵ�ȼ�շ�Ӧ�Ƿ��ȵģ������ʱ��Ǹ�ֵ���������Ϊ��̬����Ҫ�������������ԡ�H1����H2������ȷ��
�����ʵ�ȼ�շ�Ӧ�Ƿ��ȵģ������ʱ��Ǹ�ֵ��Һ̬ˮ��Ϊ��̬ˮ�Ĺ��������ȵģ��ʡ�H1����H2���ʴ���
��CaCO3��s���TCaO��s��+CO2��g����Ӧ���ȡ�H1��0��CaO��s��+H2O��l���TCa��OH��2��s�����ȡ�H2��0�����ԡ�H1����H2������ȷ��
��ѡD��
���� ���⿼�������ʷ�Ӧ�����仯�����жϣ���Ҫ�Ǹ�˹���ɵ�Ӧ�ã���������ͬ��״̬��ͬ�����ﲻͬ����Ӧ�������仯���⣬��Ŀ�ϼ�

��ϰ��ϵ�д�
�����Ŀ
11����Ч�ܵ�ص��з���Լ�綯�������ƹ㣮��һ�����͵�ȼ�ϵ�أ����Զ������Ϊ�缫����KOH��Һ�У�Ȼ��ֱ�������ͨ����������������ܷ�ӦʽΪ��2C2H6+7O2+8KOH�T4K2CO3+10H2O���йش˵�ص��ƶ���ȷ���ǣ�������
| A�� | ������ӦΪ��14H2O+7O2+28e-�T28OH- | |
| B�� | �ŵ������KOH�����ʵ���Ũ�Ȳ��� | |
| C�� | ÿ����1 mol C2H6�����·��ת�Ƶĵ���Ϊ14 mol | |
| D�� | �ŵ�һ��ʱ�������Χ�ļ�����ǿ |
8�������й�˵�����ʾ��ȷ���ǣ�������
| A�� | ���ۺ���ά�ض����ã�C6H10O5��n��ʾ������Ϊͬ���칹�� | |
| B�� |  �ȿɱ�ʾ18O2-��Ҳ���Ա�ʾ16O2- �ȿɱ�ʾ18O2-��Ҳ���Ա�ʾ16O2- | |
| C�� | Fe-2e-�TFe2+�ɱ�ʾ�������ⸯʴ��������ʴ��������Ӧ | |
| D�� | �Ҵ����ӵı���ģ�ͣ� |
15������������ͬ���칹�壨�����������칹�������ǣ�������
| A�� | �Ҵ� | B�� | ���� | C�� | ���ȱ��� | D�� | ��ϩ |
5�����ж���ijЩ���ӣ������ʣ��ļ��鼰����һ����ȷ���ǣ�������
| A�� | ����̼������Һ������ɫ�������ټ����ᣬ��ɫ������ʧ����һ����Ba2+ | |
| B�� | �����Ȼ�����Һ�а�ɫ�����������ټ����ᣬ��������ʧ��һ����SO42- | |
| C�� | ����ϡ������ټ�����������������ɫ��������ԭ��Һһ����Cl- | |
| D�� | ����ϡ���������ɫ���壬ԭ����һ����CO32- |
12�����������ʵıȽ��У���ȷ���ǣ�������
| A�� | ���뾶��O2-��F-��Na+��Li+ | B�� | ��һ�����ܣ�He��Ne��Ar | ||
| C�� | ���ۼ��ļ��ܣ�C-C��C=C��C��C | D�� | �����еļ��ǣ�CH4��H2O��CO2 |
9���۲��������ʵĽṹ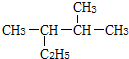 �����������Ϻ��У���������̼ԭ�ӣ�
�����������Ϻ��У���������̼ԭ�ӣ�
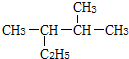 �����������Ϻ��У���������̼ԭ�ӣ�
�����������Ϻ��У���������̼ԭ�ӣ�| A�� | �ĸ� | B�� | ��� | C�� | ���� | D�� | �߸� |

