��Ŀ����
��������ͼ�е�ʵ��װ�òⶨ����ͭ������һ��Ũ�ȵ�Ũ���ᷴӦ���ɵ�NO�����NO2��������ʵ���֮�ȣ���m��ʾ��������װ��C�ɹ��ƿ����������ɣ��������ɼס���������������ɣ�����Ƥ����ͨ����װ������ˮ�����п̶ȣ�0mL��50mL�����ҹܿ������ƶ����ش��������⣺
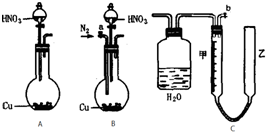
��1��ʵ�������壨NO��NO2������װ��Ӧѡ��______����װ����ţ���
��2��ʵ���е����壨NO��NO2��ͨ��Cװ�ã�������ͼ�Ĺ��ƿ�н������ܻ�����

��3��Ҫ�ⶨmֵ����Ҫ��C�е���Һ�����к͵ζ�������Ũ�������ǿ�����ԣ��ܰ����ָʾ��������ɫ��Ӱ��ʵ�飮��ˣ��к͵ζ�ǰ������еIJ�����______��
��4�����Ӻ�װ�ã���������Ժ�Ӧ����������Ҫ�IJ�������һ����b����Cװ�õ��ҹ��м�����ˮ���ڶ���______��
��5��ʵ��ǰ�ס�������Һ����ͬһˮƽ���ϣ�������ʱ�ҹܵ�Һ����ڼܵ�Һ�森��ʱӦ���еIJ�����______��
��6��ʵ���������������������������ɱ�״���£�ΪV��L����C����������ʵ���Ϊ n mol�����ú�n��V��ʽ�ӱ�ʾm=______������ɲ��ػ���

��1��ʵ�������壨NO��NO2������װ��Ӧѡ��______����װ����ţ���
��2��ʵ���е����壨NO��NO2��ͨ��Cװ�ã�������ͼ�Ĺ��ƿ�н������ܻ�����

��3��Ҫ�ⶨmֵ����Ҫ��C�е���Һ�����к͵ζ�������Ũ�������ǿ�����ԣ��ܰ����ָʾ��������ɫ��Ӱ��ʵ�飮��ˣ��к͵ζ�ǰ������еIJ�����______��
��4�����Ӻ�װ�ã���������Ժ�Ӧ����������Ҫ�IJ�������һ����b����Cװ�õ��ҹ��м�����ˮ���ڶ���______��
��5��ʵ��ǰ�ס�������Һ����ͬһˮƽ���ϣ�������ʱ�ҹܵ�Һ����ڼܵ�Һ�森��ʱӦ���еIJ�����______��
��6��ʵ���������������������������ɱ�״���£�ΪV��L����C����������ʵ���Ϊ n mol�����ú�n��V��ʽ�ӱ�ʾm=______������ɲ��ػ���
��1��������Cu�ۺ�������ŨHNO3��Ӧ����ʼӦ�к���ɫ��NO2�������ɣ�Cu+4HNO3��Ũ��=Cu��NO3��2+2H2O+2NO2�������ŷ�Ӧ�Ľ��У�HNO3�������ģ�HNO3��Ũ�����Ͷ����ϡHNO3����ʱ�䷴Ӧ�Լ������У���������������ҪΪ��ɫ��NO���壺3Cu+8HNO3��ϡ��=3Cu��NO3��2+4H2O+2NO�����������巢��װ�õ�����������Ҫ��NO��NO2��ɵĻ�����壬Ϊ�˷�ֹ���ɵ�NO�������е�O2�����������ڷ�Ӧ��ʼǰ����װ����ͨN2�����������еĿ������ʴ�Ϊ��B
��2��װ���вⶨ���������װ������������ѹǿ��ϵ�ڿ��п̶ȵļײ������ж��������������Ӧ�����ɵ����庬�ж���������һ������������������Ҫͨ��ˮ��ȥ����������������Ӧ�����̳���װ��ͼΪ��
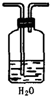
���ʴ�Ϊ

��
��3����C�е���Һ�����к͵ζ�������Ũ�������ǿ�����ԣ��ܰ����ָʾ��������ɫ��Ӱ��ʵ�飮��ˣ��к͵ζ�ǰ�����ˮϡ�ͣ�
�ʴ�Ϊ����C�е���Һ������ˮϡ�ͣ�
��4�����Ӻ�װ�ã���������Ժ�Ӧ����������Ҫ�IJ�������һ����b����Cװ�õ��ҹ��м�����ˮ��Ϊ�������ɵ�һ���������屻�����е�������������Ҫͨ�뵪�����ѿ����Ͼ���
�ʴ�Ϊ����a��ͨ����N2���ž�װ���еĿ�����
��5��ʵ��ǰ�ס�������Һ����ͬһˮƽ���ϣ�������ʱ�ҹܵ�Һ����ڼܵ�Һ�棬Ӧ�����ƶ��ҹܣ�ʹ�ס�������Һ����ͬһˮƽ���ϣ�
�ʴ�Ϊ��Ӧ�����ƶ��ҹܣ�ʹ�ס�������Һ����ͬһˮƽ���ϣ�
��6��ʵ���������������������������ɱ�״���£�ΪV��L������Ϊһ�����������ʵ���=
��C����������ʵ���Ϊ n mol���Ƕ���������ˮ��Ӧ���ɵģ���Ӧ�Ļ�ѧ����ʽΪ��3NO2+H2O=2HNO3+NO����������nmol������Ҫ���������������ʵ���Ϊ1.5nmol������һ�������������ʵ���Ϊ0.5nmol����ͭ��Ũ��ˮ��Ӧ���ɵ������У������������ʵ���Ϊ1.5nmol��һ�������������ʵ���=
mol-0.5nmol������ͭ������һ��Ũ�ȵ�Ũ���ᷴӦ���ɵ�NO�����NO2��������ʵ���֮�ȣ�ͨ������õ���
m=��
-0.5n����1.5n=��V-11.2n����33.6n��
�ʴ�Ϊ����V-11.2n����33.6n��
��2��װ���вⶨ���������װ������������ѹǿ��ϵ�ڿ��п̶ȵļײ������ж��������������Ӧ�����ɵ����庬�ж���������һ������������������Ҫͨ��ˮ��ȥ����������������Ӧ�����̳���װ��ͼΪ��

���ʴ�Ϊ

��
��3����C�е���Һ�����к͵ζ�������Ũ�������ǿ�����ԣ��ܰ����ָʾ��������ɫ��Ӱ��ʵ�飮��ˣ��к͵ζ�ǰ�����ˮϡ�ͣ�
�ʴ�Ϊ����C�е���Һ������ˮϡ�ͣ�
��4�����Ӻ�װ�ã���������Ժ�Ӧ����������Ҫ�IJ�������һ����b����Cװ�õ��ҹ��м�����ˮ��Ϊ�������ɵ�һ���������屻�����е�������������Ҫͨ�뵪�����ѿ����Ͼ���
�ʴ�Ϊ����a��ͨ����N2���ž�װ���еĿ�����
��5��ʵ��ǰ�ס�������Һ����ͬһˮƽ���ϣ�������ʱ�ҹܵ�Һ����ڼܵ�Һ�棬Ӧ�����ƶ��ҹܣ�ʹ�ס�������Һ����ͬһˮƽ���ϣ�
�ʴ�Ϊ��Ӧ�����ƶ��ҹܣ�ʹ�ס�������Һ����ͬһˮƽ���ϣ�
��6��ʵ���������������������������ɱ�״���£�ΪV��L������Ϊһ�����������ʵ���=
| VL |
| 22.4L/mol |
| VL |
| 22.4L/mol |
m=��
| VL |
| 22.4L/mol |
�ʴ�Ϊ����V-11.2n����33.6n��

��ϰ��ϵ�д�
�����Ŀ






