��Ŀ����
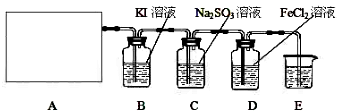
ij�о���ѧϰС��������ϵ�֪��Ư����������Һ��Ӧ����ȡ��������ѧ����ʽΪ��Ca(ClO)2+CaCl2+2H2SO4�������������ȡ��������֤�����ʵ�ʵ�飺

ѡ��_________װ�ã���дװ�õ���ţ���ȡ������

��2����������������һ��ʵ�飬֤��ϴ��ƿC�е�Na2SO3�Ѳ��ֱ�����������ʵ�鲽�裩��
____________________________________________________________________
��3��д����Dװ���з�����Ӧ�����ӷ���ʽ��______________________________________
��4����ʵ��������Ե�ȱ�ݣ���������Ľ��ķ�����
____________________________________________________________________
��5����С���ֽ���������ʵ�飺��ȡƯ��
��1��b
��2��ȡ��Һ�������ڽྻ���Թ��У������еμ�ϡ���������ٲ����������������е����Ȼ�����Һ����������ɫ��������֤�����������Ѳ��ֱ�������
��3��Cl2+H2O====H++Cl-+HClO
H++![]() =CO2��+H2O
=CO2��+H2O
��4����β��ͨ��NaOH��Һ��
��5��35.75%
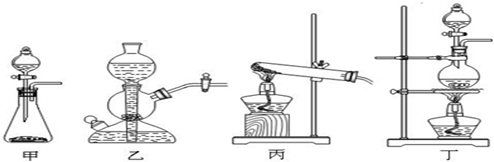
��������1��A����������ȡCl2����������Cl2�����ɷ�Ӧ�ķ�Ӧ��״̬�ͷ�Ӧ�Ƿ���Ҫ����ѡ�����巢��װ�ã�Ӧѡ���ڡ���+Һ![]() �����͵�b���䷴Ӧ�Ļ�ѧ����ʽΪMnO2+4HCl��Ũ��
�����͵�b���䷴Ӧ�Ļ�ѧ����ʽΪMnO2+4HCl��Ũ��![]() MnCl2+Cl2��+2H2O����2��Na2SO3�����IJ���ΪNa2SO4,ȷ������
MnCl2+Cl2��+2H2O����2��Na2SO3�����IJ���ΪNa2SO4,ȷ������![]() ����֤��Na2SO3�Ѳ��ֱ�����������ʱ��ӦȡC����Һ�����ڽྻ�Թ��У��ȼ�ϡHCl�����ٲ����������ų�
����֤��Na2SO3�Ѳ��ֱ�����������ʱ��ӦȡC����Һ�����ڽྻ�Թ��У��ȼ�ϡHCl�����ٲ����������ų�![]() �ĸ��ţ�Ȼ���ټ���BaCl2��Һ����������ɫ��������֤��Na2SO3�Ѳ��ֱ���������3������D��Cl2��ˮ��Ӧ�����ӷ���ʽΪCl2+H2O
�ĸ��ţ�Ȼ���ټ���BaCl2��Һ����������ɫ��������֤��Na2SO3�Ѳ��ֱ���������3������D��Cl2��ˮ��Ӧ�����ӷ���ʽΪCl2+H2O![]() H++Cl-+HClO,HCl����NaHCO3��Ӧ�����ӷ���ʽΪH++
H++Cl-+HClO,HCl����NaHCO3��Ӧ�����ӷ���ʽΪH++![]() ====CO2��+H2O��HClO�����Ա�H2CO3����������NaHCO3��Ӧ����4��NaHCO3����Cl2���������ޣ�Ӧ��β��ͨ��NaOH��Һ�С���5����ϵʽCa(ClO)2��2Cl2��2I2��4Na2S2O3,n��Ca(ClO)2��=
====CO2��+H2O��HClO�����Ա�H2CO3����������NaHCO3��Ӧ����4��NaHCO3����Cl2���������ޣ�Ӧ��β��ͨ��NaOH��Һ�С���5����ϵʽCa(ClO)2��2Cl2��2I2��4Na2S2O3,n��Ca(ClO)2��=![]() n(Na2S2O3)=20.0 mL��10
n(Na2S2O3)=20.0 mL��10![]() ��
��![]() =0.005 mol,Ca(ClO)2%=
=0.005 mol,Ca(ClO)2%=![]() ��100%=35.75%��
��100%=35.75%��

 �¿α�ͬ��ѵ��ϵ�д�
�¿α�ͬ��ѵ��ϵ�д� һ����ʦ����Ӧ����������һ��ȫϵ�д�
һ����ʦ����Ӧ����������һ��ȫϵ�д�