��Ŀ����
��14�֣���������һ����Ҫ�����ȼ�ϣ�Ҳ�������������������ȣ��Գ��������ƻ����á���ҵ�Ͽ�����ú���������ˮú�����ϳɶ����ѡ���ش��������⣺
�� ú����������Ҫ��ѧ��Ӧ����ʽΪ��___________________________��
�� ����ˮú���ϳɶ����ѵ�������Ӧ���£�
�� 2H2(g)
+ CO(g) CH3OH(g)
��H����90.8 kJ��mol��1 K1
CH3OH(g)
��H����90.8 kJ��mol��1 K1
�� 2CH3OH(g) CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1 K2
CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1 K2
�� CO(g) + H2O(g) CO2(g)
+ H2(g)����H����41.3 kJ��mol��1 K3
CO2(g)
+ H2(g)����H����41.3 kJ��mol��1 K3
�ܷ�Ӧ��3H2(g) +
3CO(g) CH3OCH3(g) +CO2 (g)����H�� _____ , K=
���ð���K1�� K2�� K3��ʽ�ӱ�ʾ��.һ�������µĵ����ܱ������У����ܷ�Ӧ�ﵽƽ�⣬Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ��
������ĸ���ţ���
CH3OCH3(g) +CO2 (g)����H�� _____ , K=
���ð���K1�� K2�� K3��ʽ�ӱ�ʾ��.һ�������µĵ����ܱ������У����ܷ�Ӧ�ﵽƽ�⣬Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ��
������ĸ���ţ���
a�����¸�ѹ b��������� c����CO2��Ũ��
d������CO��Ũ�� e������H2��Ũ�� f������He
��3����֪��Ӧ��2CH3OH(g)
 CH3OCH3(g) + H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400 �����¶��£����ܱ������м���CH3OH ����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
CH3OCH3(g) + H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400 �����¶��£����ܱ������м���CH3OH ����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
|
���� |
CH3OH |
CH3OCH3 |
H2O |
|
Ũ��/��mol��L��1�� |
0.44 |
0.6 |
0.6 |
�� �Ƚϴ�ʱ�����淴Ӧ���ʵĴ�С��v������ ______ v���棩 ���>������<������)��
�� ������CH3OH��10 min��Ӧ�ﵽƽ�⣬��ʱc(CH3OH) �� _________����ʱ���ڷ�Ӧ����v(CH3OH) �� __________��
(1) C+H2O CO+H2 (2)
-246.4kJ�� mol -1��K12��K2�� K3
��c��e
CO+H2 (2)
-246.4kJ�� mol -1��K12��K2�� K3
��c��e
(3) �٣� ��0.04 mol��L-1 ��0.16 mol��L-1��min-1��ÿ��2�֣�
����������1��ú������Ҫ���ڸ�����ú��ˮ������Ӧ�ϳ�ˮú�������Է���ʽΪ
C+H2O CO+H2��
CO+H2��
��2�������˹���ɵ�Ӧ�ú�ƽ�ⳣ���ļ��㡣�١�2���ڣ��ۼ��õ�
3H2(g)
+ 3CO(g) CH3OCH3(g) +CO2 (g)��������H���ڣ���90.8��2��23.5��41.3��kJ��mol��1 ����246.4kJ��
mol -1�����ݢ١�2���ڣ��ۿɼ���ƽ�ⳣ��ΪK��K12��K2�� K3����Ϊ��Ӧ�������С�ġ����ȵĿ��淴Ӧ���������淴Ӧ�����ƶ����������ܸı�ƽ��״̬������������Ũ��ƽ��������Ӧ�����ƶ�������CO��Ũ��ƽ��������Ӧ�����ƶ�������������ת���ʣ���������Ũ�ȿ������CO��ת���ʣ�����������䣬����ϡ�����壬ƽ�ⲻ�ƶ�����˴���ce��
CH3OCH3(g) +CO2 (g)��������H���ڣ���90.8��2��23.5��41.3��kJ��mol��1 ����246.4kJ��
mol -1�����ݢ١�2���ڣ��ۿɼ���ƽ�ⳣ��ΪK��K12��K2�� K3����Ϊ��Ӧ�������С�ġ����ȵĿ��淴Ӧ���������淴Ӧ�����ƶ����������ܸı�ƽ��״̬������������Ũ��ƽ��������Ӧ�����ƶ�������CO��Ũ��ƽ��������Ӧ�����ƶ�������������ת���ʣ���������Ũ�ȿ������CO��ת���ʣ�����������䣬����ϡ�����壬ƽ�ⲻ�ƶ�����˴���ce��
��3���������ݿɼ����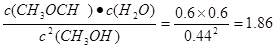 ��С��ƽ�ⳣ�����������Ӧ���ʴ��淴Ӧ���ʡ���Ϊ�������ļ״���1.64mol��L��1�����Ը��ݷ�Ӧʽ�ɽ��м��㡣
��С��ƽ�ⳣ�����������Ӧ���ʴ��淴Ӧ���ʡ���Ϊ�������ļ״���1.64mol��L��1�����Ը��ݷ�Ӧʽ�ɽ��м��㡣
2CH3OH(g)  CH3OCH3(g) + H2O(g)
CH3OCH3(g) + H2O(g)
��ʼŨ�ȣ�mol��L��1�� 1.64 0 0
ת��Ũ�ȣ�mol��L��1�� x 0.5x 0.5x
ƽ��Ũ�ȣ�mol��L��1�� 1.64��x 0.5x 0.5x
����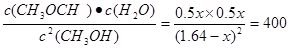
���x��1.6������ƽ��ʱc(CH3OH)����0.04mol��L��1����Ӧ����v(CH3OH) ����0.16 mol��L-1��min-1��

3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g����H=-274kJ?mol-1��
��1���÷�Ӧ��һ�������µ��ܱ������дﵽƽ���Ϊͬʱ��߷�Ӧ���ʺͶ����ѵIJ��ʣ����Բ�ȡ�Ĵ�ʩ��
A�������¶� B��������� C����С������� D������H2��Ũ�� E�������������
��2���÷�Ӧ���Է��������У�
4H2��g��+2CO��g���TCH3OCH3��g��+H2O��g����H1 ��
CO��g��+H2O��g���TCO2��g��+H2��H2=-42kJ?mol-1
��Ӧ�ٵ��ʱ��H1=
��֪ijЩ��ѧ���ļ����������±���ʾ��
| ��ѧ�� | C-H | O-H | H-H | C-O | C��O |
| ����/kJ��mol-1 | 413 | 463 | 436 | 358 | 1072 |
��3��������Ҳ����ͨ��CH3OH���Ӽ���ˮ�Ƶã�
2CH3OH��g��?CH3OCH3��g��+H2O��g����H3=-23.5kJ?mol-1����T1�棬�����ܱ������н�������ƽ�⣬��ϵ�и����Ũ�ȱ仯���±���
| ���� | CH3OH ��g�� | CH3OCH3 ��g�� | H2O ��g�� |
| ��ʼŨ��/mol��l-1 | 2 | 0.2 | 0 |
| ƽ��Ũ��/mol��l-1 | 0.4 | 1 | 0.8 |
����ͬ�����£����ı���ʼŨ�ȣ�
| ���� | CH3OH ��g�� | CH3OCH3 ��g�� | H2O ��g�� |
| ��ʼŨ��/mol��l-1 | 0.4 | 1.2 | 0.6 |
��4��������-������һ�ָ�Ч��ȼ�ϵ�أ�������������Һ�У���ط�Ӧ����ʽΪ��
CH3OCH3��l��+3O2��g��+4KOH��aq���T2K2CO3��aq��+5H2O��l��
��д�������ĵ缫��Ӧʽ��
 ��2012?�������ģ����������һ����Ҫ�����ȼ�ϣ�Ҳ�������������������ȣ��Գ��������ƻ����ã���ҵ������H2��CO2�ϳɶ����ѵķ�Ӧ���£�6H2��g��+2CO2��g��?CH3OCH3��g��+3H2O��g��
��2012?�������ģ����������һ����Ҫ�����ȼ�ϣ�Ҳ�������������������ȣ��Գ��������ƻ����ã���ҵ������H2��CO2�ϳɶ����ѵķ�Ӧ���£�6H2��g��+2CO2��g��?CH3OCH3��g��+3H2O��g��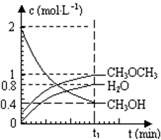 ��֪��������һ����Ҫ�����ȼ�ϣ�����ͨ��CH3OH���Ӽ���ˮ�Ƶã�2CH3OH��g��?CH3OCH3��g��+H2O��g����H=23.5kJ?mol-1����T1�棬�����ܱ������н�������ƽ�⣬��ϵ�и����Ũ����ʱ��仯��ͼ��ʾ����ش��������⣺
��֪��������һ����Ҫ�����ȼ�ϣ�����ͨ��CH3OH���Ӽ���ˮ�Ƶã�2CH3OH��g��?CH3OCH3��g��+H2O��g����H=23.5kJ?mol-1����T1�棬�����ܱ������н�������ƽ�⣬��ϵ�и����Ũ����ʱ��仯��ͼ��ʾ����ش��������⣺