��Ŀ����
ij�о���ѧϰС������Na2O2��ˮ��Ӧʵ��ʱ������Na2O2��ˮ��Ӧ�����Һ�еμӷ�̪��Һ���ֺ�ɫ������ɫ�ܿ���ȥ���ס��ҡ�����ͬѧ�Դ�����ֱ����������Ʋ⣺
�ף���Ϊ��Ӧ���Թܺ��ȣ����Կ�������Һ�¶Ƚϸ�ʹ��ɫ��ȥ��
�ң���Ϊ����ˮ�������٣���ɫ��ȥ���������ɵ�NaOH��ҺŨ�Ƚϴ��Ӱ�졣
����Na2O2����ǿ�����ԣ���������O2��H2O2�����ܲ����Ҳ����ǿ�����ԣ�����������Ư���˺�ɫ���ʡ�
��1����֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ�����_______________����֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ�����_______________��������________����ʱ������˵����ͬѧ���Ʋ���ȷ��
��2����ͬѧ����ö����ķ���̽����ͬѧ�������Ƿ���H2O2����ʵ�鷽��Ϊ����ȡ
2.6 g Na2O2���壬ʹ֮��������ˮ��Ӧ����������O2�������������ֵ�Ƚϣ����ɵó����ۡ�
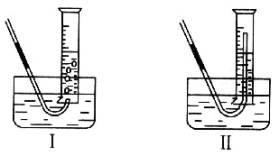
�������������ʱ��������Թܺ���Ͳ�ڵ����嶼��ȴ������ʱ���У�Ӧѡ����ͼװ���е�________��������___________��
�����ڱ�״���²�������������Ӧѡ�õ���Ͳ�Ĵ�С���Ϊ______��ѡ�100 m L������200 m L������500 m L����1000 m L������
������
��1����ȴ��ӷ�̪��Һ����ȴ����Һ�Ƿ��죻��ˮϡ�ͺ���Һ�Ƿ��죨��ӽ϶��ˮ��Ӧ��μӷ�̪��Һ���Ƿ��죩����ȴ��ˮϡ�ͺ������졣 ��2����II����ֹ������ȴʱ��������������500 m L��
|
��ʾ��
��1������Ϊ��ɫ��ȥ��ԭ������Һ���¶Ƚϸߣ���˽���Һ��ȴ����Һ�Ƿ��죬��ԭ��Һ��ȴ���ټӷ�̪���Ƿ��죬�Կ��ж����Ƿ���ȷ������Ϊ��ɫ��ȥ��ԭ����NaOH��Ũ�ȴ���ˣ���������Һϡ�Ϳ��Ƿ��죬�����϶��ˮ��ַ�Ӧ���ٵμӷ�̪����Һ�Ƿ���Կ���֤�ҵķ����Ƿ���ȷ����������������Һ�Ա��ɫ����ͬʱ֤�������ƶ�����ȷ�ġ� ��2������ȴ����������������С�����ײ�����������Ϊ��ֹ������ķ�������Ӧѡ��װ�ã�II������2.6 g Na2O2��H2O��Ӧ������O2�����ΪV��O2�� 2Na2O2 + 2H2O == 4NaOH + O2�� 156 g 22400 m L 2.6 g V(O2)
Ӧѡ��500 m L����Ͳ��
|


�ף���Ϊ������NaOH��Һ��Ӧ��ʹ��Һ���Լ������������ԣ�������Һ��ɫ��ȥ���ң���Ϊ��������ˮ����ˮ��Ӧ����HClO������HClO������Ư�����ö�ʹ��Һ��ɫ��?
��1����֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ����ǣ�________________________________��?
��֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ����ǣ�________________________��?
��2�������NaOH��Һ�ij�KMnO4������Һ����Cl2����SO2���壬������ͼʾ����װ�������Ȫʵ���Ƿ����?________(���������������)��?
�����Ϊ����������˵���������________________________________________________________��?
��3�����ơ���ɫ��Ȫʵ�顱���ж��֡���ɫ����Ȫʵ�顱�����㰴ʵ��Ҫ�ֱ����1������ɫ��Ȫʵ�顱����д���б���
��� | ʵ��Ҫ�� | �ձ��е���Һ | �ι��е�Һ�� | ��ƿ�е����� |
�� | ��ɫ���ɫ��Ȫ |
| H2O |
|
�� | ��ɫ����ɫ��Ȫ |
|
| SO2 |

�ף���Ϊ������NaOH��Һ��Ӧ��ʹ��Һ���Լ������������ԣ�������Һ��ɫ��ȥ���ң���Ϊ��������ˮ����ˮ��Ӧ����HClO������HClO������Ư�����ö�ʹ��Һ��ɫ��
(1)��֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ����ǣ�____________________��
��֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ����ǣ�______________________��
(2)�����NaOH��Һ�ij�KMnO4������Һ����Cl2����SO2���壬������ͼʾ����װ�������Ȫʵ���Ƿ����? _____________(���������������)�������Ϊ����������˵���������______________________________��
(3)���ơ���ɫ��Ȫʵ�顱���ж��֡���ɫ����Ȫʵ�顱�����㰴ʵ��Ҫ�ֱ����1������ɫ��Ȫʵ�顱����д���б���
��� | ʵ��Ҫ�� | �ձ��е���Һ | �ι��е�Һ�� | ��ƿ�е����� |
�� | ��ɫ���ɫ��Ȫ |
| H2O |
|
�� | ��ɫ����ɫ��Ȫ |
|
| SO2 |

ͼ1-5-30
�ף���Ϊ������NaOH��Һ��Ӧ��ʹ��Һ���Լ������������ԣ�������Һ��ɫ��ȥ��
�ң���Ϊ��������ˮ����ˮ��Ӧ����HClO������HClO������Ư�����ö�ʹ��Һ��ɫ��
(1)��֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ����ǣ���֤��ͬѧ���Ʋ��Ƿ���ȷ�ķ�����_____________________________________________________________________��
(2)�����NaOH��Һ�ij�����KMnO4��Һ����Cl2����SO2���壬������ͼʾ����װ�������Ȫʵ���Ƿ������__________ (���������������)�������Ϊ����������˵��������ɣ�_____________________________________________________________________��
(3)���ơ���ɫ��Ȫʵ�顱���ж��֡���ɫ����Ȫʵ�顱�����㰴ʵ��Ҫ�ֱ����1������ɫ��Ȫʵ�顱������д���б���
��� | ʵ��Ҫ�� | �ձ��е���Һ | �ι��е�Һ�� | ��ƿ�е����� |
�� | ��ɫ���ɫ����Ȫ |
| H2O |
|
�� | ��ɫ����ɫ����Ȫ |
|
| SO2 |
(4)���������������Ȫʵ���װ��(��ͼ1-5-31��ʾ����ͷ�������㹻���Ľ�����)����ԭ����_______________��ʵ������з��ֿ�ʼʱ�������Ȫѹ����ˮ�����������Խ��Խ����������ƿ���ܳ�����Һ���������㹻���ģ�װ��Ҳ��©������˵��ԭ��________________
____________________________________________________________________��

ͼ1-5-31