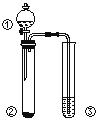
جâؤ؟ؤعبف
،¾جâؤ؟،؟ؤ³·د´ك»¯¼ء؛¬58.2%µؤSiO2،¢21.0%µؤZnO،¢4.5%µؤZnS؛ح12.8%µؤCuS،£ؤ³ح¬ر§سأ15.0 g¸أ·د´ك»¯¼ءخھشءد£¬»طتصئنضذµؤذ؟؛حح،£²ةسأµؤتµرé·½°¸بçدآ£؛

»ط´ًدآءذختجâ£؛
£¨1£©شعدآءذ×°ضأضذ£¬µعز»´خ½³ِ±طذëسأ____________£¬µع¶´خ½³ِس¦ر،سأ____________،££¨جî±ê؛إ£©

£¨2£©µع¶´خ½³ِت±£¬دٍت¢سذآثز؛1µؤ·´س¦ئ÷ضذ¼سبëد،ءٍثل£¬؛َµخبë¹رُ»¯اâبـز؛،£بôث³ذٍدà·´£¬»لشى³ة___________،£آثشü2µؤض÷زھ³ة·ضتا____________________،£
£¨3£©إ¨ثُءٍثلذ؟،¢ءٍثلحبـز؛ت¹سأµؤئ÷أَأû³ئتا________________،£
£¨4£©ؤ³ح¬ر§شعتµرéحê³ةض®؛َ£¬µأµ½1.5gCuSO4©q5H2O,شٍحµؤ»طتصآتخھ__________________،£
،¾´ً°¸،؟£¨1£©D A £¨أ؟؟ص1·ض£¬¹²2·ض£©
£¨2£©H2O2سë¹ججه؟إء£½س´¥·ض½â£¨أ؟؟ص2·ض£¬¹²4·ض£©
£¨3£©صô·¢أَ£¨2·ض£©
£¨4£©30% £¨3·ض£©
،¾½âخِ،؟
تشج⣨1£©¸ù¾فجâ¸ّ»¯ر§¹¤زصء÷³جضھµعز»´خ½³ِ·¢ةْ·´س¦ZnO+H2SO4![]() ZnSO4+H2O،¢ZnS+H2SO4
ZnSO4+H2O،¢ZnS+H2SO4![]() ZnSO4+H2S£¬سذسذ¶¾ئّجهH2Sةْ³ة£¬±طذëسأاâرُ»¯ؤئبـز؛½ّذذخ²ئّ´¦ہي£¬ر،D×°ضأ£¬µع¶´خ½³ِت±·¢ةْ·´س¦£؛CuS+H2O2+H2SO4
ZnSO4+H2S£¬سذسذ¶¾ئّجهH2Sةْ³ة£¬±طذëسأاâرُ»¯ؤئبـز؛½ّذذخ²ئّ´¦ہي£¬ر،D×°ضأ£¬µع¶´خ½³ِت±·¢ةْ·´س¦£؛CuS+H2O2+H2SO4![]() CuSO4+S+2H2O£¬²»²ْةْسذ¶¾ئّجه£¬؟ةر،سأA×°ضأ،£
CuSO4+S+2H2O£¬²»²ْةْسذ¶¾ئّجه£¬؟ةر،سأA×°ضأ،£
£¨2£©µع¶´خ½³ِت±£¬دٍت¢سذآثز؛1µؤ·´س¦ئ÷ضذ¼سبëد،ءٍثل£¬؛َµخبë¹رُ»¯اâبـز؛،£بôث³ذٍدà·´£¬»لشى³ةH2O2سë¹ججه؟إء£½س´¥·ض½â،£آثشü2µؤض÷زھ³ة·ضتا¶رُ»¯¹è،£
£¨3£©إ¨ثُءٍثلذ؟،¢ءٍثلحبـز؛ت¹سأµؤئ÷أَأû³ئتاصô·¢أَ،£
£¨4£©15.0 g·د´ك»¯¼ءضذ؛¬سذحµؤخïضتµؤء؟خھ15.0g،ء12.8%،آ96g/mol=0.02mol£¬1.5gCuSO4©q5H2Oضذحµؤخïضتµؤء؟خھ1.5g،آ250g/mol=0.006mol£¬شٍحµؤ»طتصآتخھ0.006mol/0.02mol،ء100%=30%،£

 ذ،ر§؟خت±جطرµدµءذ´ً°¸
ذ،ر§؟خت±جطرµدµءذ´ً°¸،¾جâؤ؟،؟KAl£¨SO4£©2،¤12H2O£¨أ÷·¯£©تاز»ضض¸´رخ£¬شعشىض½µب·½أوس¦سأ¹م·؛،£تµرéتزضذ£¬²ةسأ·دز×ہ¹ق£¨ض÷زھ³ة·ضخھAl£¬؛¬سذةظء؟µؤFe،¢Mgشسضت£©ضئ±¸أ÷·¯µؤ¹³جبçدآح¼ثùت¾،£»ط´ًدآءذختجâ£؛
![]()
£¨1£©خھ¾،ء؟ةظزبëشسضت£¬تش¼ء¢ظس¦ر،سأ___£¨جî±ê؛إ£©،£
A£®HClبـز؛ | B£®H2SO4بـز؛ | C£®°±ث® | D£®NaOHبـز؛ |
£¨2£©ز×ہ¹قبـ½â¹³جضذض÷زھ·´س¦µؤ»¯ر§·½³جت½خھ________،£
£¨3£©³ءµيBµؤ»¯ر§ت½خھ________£»½«ةظء؟أ÷·¯بـسعث®£¬بـز؛³تبُثلذش£¬ئنشزٍتا__________،£
£¨4£©زرضھ£؛Kw=1.0،ء10-14£¬Al£¨OH£©3![]() AlO2-+H++H2O K=2.0،ء10-13،£Al(OH)3بـسعNaOHبـز؛·´س¦µؤئ½؛â³£تµبسع_________،£
AlO2-+H++H2O K=2.0،ء10-13،£Al(OH)3بـسعNaOHبـز؛·´س¦µؤئ½؛â³£تµبسع_________،£
،¾جâؤ؟،؟دضت¹سأثل¼îضذ؛حµخ¶¨·¨²â¶¨تذتغ°×´×µؤ×ـثلء؟(g/100 mL)،£
¢ٌ.تµرé²½ضè£؛إنضئ´²â°×´×بـز؛£¬سأثلت½µخ¶¨¹ـء؟ب،10.00 mLت³سأ°×´×£¬شعةص±ضذسأث®د،تح؛َ×ھزئµ½100 mLبفء؟ئ؟ضذ¶¨بف£¬ز،شب¼´µأ´²â°×´×بـز؛،£ء؟ب،´²â°×´×بـز؛20.00mLسع׶ذخئ؟ضذ£¬دٍئنضذµخ¼س2µخ·سجھ×÷ض¸ت¾¼ء،£
£¨1£©¶ءب،ت¢×°0.1000 mol/L NaOHبـز؛µؤ¼îت½µخ¶¨¹ـµؤ³ُت¼¶ءت،£بç¹ûز؛أوخ»ضأبçح¼ثùت¾£¬شٍ´ثت±µؤ¶ءتخھ________ mL،£
![]()
£¨2£©µخ¶¨،£إذ¶دµخ¶¨ضصµمµؤدضدَتا________________________________£¬´ïµ½µخ¶¨ضصµم£¬ح£ض¹µخ¶¨£¬²¢¼اآ¼NaOHبـز؛µؤ×îضص¶ءت،£ضط¸´µخ¶¨3´خ،£
¢ٍ.تµرé¼اآ¼£؛
µخ¶¨´خت تµرéت¾ف(mL) | 1 | 2 | 3 | 4 |
V(رùئ·) | 20.00 | 20.00 | 20.00 | 20.00 |
V(NaOH)(دû؛ؤ) | 15.95 | 15.00 | 15.05 | 14.95 |
¢َ.ت¾ف´¦ہيسëجضآغ£؛
£¨3£©ؤ³ح¬ر§شع´¦ہيت¾فت±¼ئثمµأ£؛ئ½¾ùدû؛ؤµؤNaOHبـز؛µؤجه»V£½(15.95£«15.00£«15.05£«14.95)/4£½15.24 mL،£ض¸³ِثûµؤ¼ئثمµؤ²»؛دہيض®´¦£؛_________________،£
£¨4£©°´صب·ت¾ف´¦ہي£¬µأ³ِc£¨تذتغ°×´×£©=_______mol،¤L-1£¬تذتغ°×´××ـثلء؟=____g،¤100mL-1،£
،¾جâؤ؟،؟ہûسأبçح¼ثùت¾×°ضأ½ّذذدآءذتµر飬ؤـµأ³ِدàس¦تµرé½لآغµؤتا
ر،دî | ¢ظ | ¢ع | ¢غ | تµرé½لآغ |
|
A | إ¨رخثل | Na2CO3 | Na2SiO3بـز؛ | ·ا½ًتôذش£؛ C>Si | |
B | ت³رخث® | µçت¯ | نهث® | ززب²؟ةسëنه·¢ةْ¼س³ة·´س¦ | |
C | إ¨دُثل | Al | NaOHبـز؛ | آء؛حإ¨دُثل·´س¦؟ةةْ³ةNO2 | |
D | إ¨ءٍثل | Na2SO3 | KMnO4ثلذشبـز؛ | SO2¾كسذ»¹شذش |
A. A B. B C. C D. D
،¾جâؤ؟،؟½üؤêہ´£¬رذ¾؟بثش±جل³ِہûسأ؛¬ءٍخïضتبب»¯ر§ر»·تµدضج«رôؤـµؤ×ھ»¯سë´و´¢£¬¹³جبçدآ£؛

£¨1£©·´س¦¢ٌ£؛2H2SO4(l)=2SO2(g)+2H2O(g)+O2(g) ،÷H1=+551kJmol-1
·´س¦¢َ£؛S(g)+O2(g)=SO2(g) ،÷H3=-297kJmol-1
·´س¦¢ٍµؤبب»¯ر§·½³جت½£؛____________________،£
£¨2£©¶ش·´س¦¢ٍ£¬شعؤ³ز»ح¶ءد±بت±£¬ء½ضضر¹ا؟دآ£¬H2SO4شعئ½؛âجهدµضذخïضتµؤء؟·ضتثوخآ¶بµؤ±ن»¯¹طدµبçسزح¼ثùت¾£¬p2______p1£¨جî،°£¾،±»ٍ،°£¼،±£©£¬µأ³ِ¸أ½لآغµؤہيسةتا__________،£

£¨3£©I-؟ةزش×÷خھث®بـز؛ضذSO2ئ绯·´س¦µؤ´ك»¯¼ء£¬؟ةؤـµؤ´ك»¯¹³جبçدآ£¬½«ii²¹³نحêصû
i£®SO2+4I-+4H+=S،+2I2+2H2O
ii£®I2+2H2O+______=______+______+2I-
£¨4£©ج½¾؟i،¢ii·´س¦ثظآتسëSO2ئ绯·´س¦ثظآتµؤ¹طدµ£¬تµرéبçدآ£؛·ض±ً½«18 mLSO2±¥؛حبـز؛¼سبëµ½2mLدآءذتش¼ءضذ£¬أـ±ص·إضأ¹غ²ىدضدَ£¬£¨زرضھ£؛I2ز×بـ½âشعKIبـز؛ضذ£©
ذٍ؛إ | A | B | C | D |
تش¼ء×é³ة | 0.4molL-1 KI | amolL-1 KI 0.2molL-1H2SO4 | 0.2molL-1H2SO4 | 0.2molL-1 KI 0.0002 molI2 |
تµرéدضدَ | بـز؛±ن»ئ£¬ز»¶خت±¼ن؛َ³ِدض»ë×ا | بـز؛±ن»ئ£¬³ِدض»ë×ا½دA؟ى | خقأ÷دشدضدَدَ | بـز؛سة×ط؛ضة«؛ـ؟ىحتة«£¬±ن³ة»ئة«£¬³ِدض»ë×ا½دA؟ى |
¢ظ BتاAµؤ¶ش±بتµر飬شٍa=______،£
¢ع±ب½دA،¢B،¢C£¬؟ةµأ³ِµؤ½لآغتا______،£
¢غتµرé±يأ÷£¬SO2µؤئ绯·´س¦ثظآتD£¾A£®½ل؛دi£¬ii·´س¦ثظآت½âتحشزٍ£؛______،£