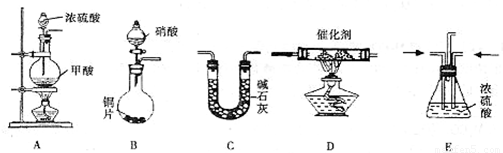
��Ŀ����
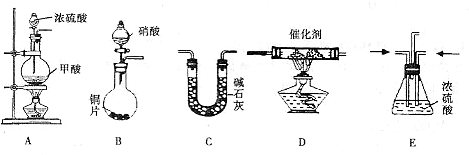
����β������Ҫ�ɷ�ΪCO2��CO�� NOx(NO��NO2�Ļ�������NO���������ռ95%����)�ȡ���������֮һ���������������ϼ�װ����ת�����������ô���ʹCO��NOx������Ӧ�� ����ת��ΪCO2��N2��ijС����ʵ����������ͼ��ʾװ��ģ������β��������CO��NOx�ķ�Ӧ����������Ӧ�����������ɡ�(��֪HCOOH CO��+H2O) �Իش��������⣺
CO��+H2O) �Իش��������⣺
 CO��+H2O) �Իش��������⣺
CO��+H2O) �Իش��������⣺
(1)����������˳��Ϊ ( )��( )��( )���ռ�����
( )��( )��( )���ռ�����
(2)Eװ�õ�������________��________��
(3)д��D��NOx��CO��Ӧ�Ļ�ѧ����ʽ________��
(4)��B���������ɵ�NOxΪNO��д���÷�Ӧ�Ļ�ѧ����ʽ�����������ת�Ƶķ������Ŀ___________��
(5)��ͨ���NOxΪNO��Cװ������8.8 g���ռ����������ڱ�״����Ϊ 4. 48 L������Է�������Ϊ28.4�������ռ�����������NO�����ʵ���Ϊ____________��
(6)ѡ���Ч������������β��ת��Ϊ�����壬�㳹���������β���Ի� ����Ӱ�죬����˵���Ƿ���ȷ���������ɣ�______________��
 ( )��( )��( )���ռ�����
( )��( )��( )���ռ�����(2)Eװ�õ�������________��________��
(3)д��D��NOx��CO��Ӧ�Ļ�ѧ����ʽ________��
(4)��B���������ɵ�NOxΪNO��д���÷�Ӧ�Ļ�ѧ����ʽ�����������ת�Ƶķ������Ŀ___________��
(5)��ͨ���NOxΪNO��Cװ������8.8 g���ռ����������ڱ�״����Ϊ 4. 48 L������Է�������Ϊ28.4�������ռ�����������NO�����ʵ���Ϊ____________��
(6)ѡ���Ч������������β��ת��Ϊ�����壬�㳹���������β���Ի� ����Ӱ�죬����˵���Ƿ���ȷ���������ɣ�______________��
(1)(E)��(D)��(C)�ռ�����
(2)���������� ��ͨ���۲����ݣ�������������
(3)2NOx+2xCO N2 +2xCO2
N2 +2xCO2
(4)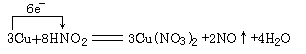
(5)0. 04 mol
(6)����ȷ���ٸ÷�ӦΪ���淴Ӧ�����ܷ�Ӧ��ȫ��
��û�ﵽ����̼�ŷš���Ŀ�ģ�������ı仯����DZ�ڵ�Ӱ���Σ����
(2)���������� ��ͨ���۲����ݣ�������������
(3)2NOx+2xCO
 N2 +2xCO2
N2 +2xCO2(4)
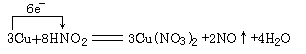
(5)0. 04 mol
(6)����ȷ���ٸ÷�ӦΪ���淴Ӧ�����ܷ�Ӧ��ȫ��
��û�ﵽ����̼�ŷš���Ŀ�ģ�������ı仯����DZ�ڵ�Ӱ���Σ����

��ϰ��ϵ�д�
�����Ŀ
 �������������������������������ռ�����
�������������������������������ռ�����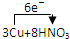 =3Cu��NO3��2+2NO��+4H2O
=3Cu��NO3��2+2NO��+4H2O
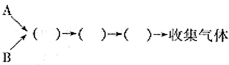


 �ռ����壨��дװ�ñ�ţ�
�ռ����壨��дװ�ñ�ţ� ��
��

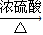 CO��+H2O��
CO��+H2O��