��Ŀ����
 ��2007?��ͷ��ģ��2012��6��16���ҹ��ɹ������ˡ����ݾźš������־���й��˵�̫��ʱ����ǰ����һ�����䡰��š�ʱ���£�N2H4����Ϊ�����������ȼ�ϣ�NO2Ϊ����������Ӧ����N2��ˮ��������֪��
��2007?��ͷ��ģ��2012��6��16���ҹ��ɹ������ˡ����ݾźš������־���й��˵�̫��ʱ����ǰ����һ�����䡰��š�ʱ���£�N2H4����Ϊ�����������ȼ�ϣ�NO2Ϊ����������Ӧ����N2��ˮ��������֪��N2��g��+2O2��g��=2NO2��g������H=+67.7kJ/mol
N2H4��g��+O2��g��=N2��g��+2H2O��g������H=-534kJ/mol
���й����º�NO2��Ӧ���Ȼ�ѧ����ʽ�У���ȷ���ǣ�������
���������ݸ�˹���ɣ�����֪�Ȼ�ѧ����ʽ���Ժ��ʵ�ϵ�����мӼ�����Ŀ���Ȼ�ѧ����ʽ����Ӧ��Ҳ������Ӧ��ϵ��������Ӧ�����㣮
����⣺��֪����N2��g��+2O2��g��=2NO2��g������H=+67.7kJ/mol
��N2H4��g��+O2��g��=N2��g��+2H2O��g������H=-534kJ/mol
���ݸ�˹���ɣ��ڡ�2-�ٵ�2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g�����ʡ�H=2����-534kJ/��-67.7kJ/mol=-1135.7 kJ/mol��
�º�NO2����N2��ˮ�������Ȼ�ѧ����ʽΪ��2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g������H=-1135.7 kJ/mol��
A��ˮ��״̬ӦΪ��̬������Һ̬����A����
B����Ӧ��Ϊ��H=-1135.7 kJ/mol�����ǡ�H=-1000.3 kJ/mol����B����
C��1molN2H4��g����Ӧ��ˮ��״̬ӦΪ��̬������Һ̬����Ӧ��Ϊ��H=-567.86 kJ/mol����C����
D��������������֪���º�NO2����N2��ˮ�������Ȼ�ѧ����ʽΪ��2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g������H=-1135.7 kJ/mol����D��ȷ��
��ѡD��
��N2H4��g��+O2��g��=N2��g��+2H2O��g������H=-534kJ/mol
���ݸ�˹���ɣ��ڡ�2-�ٵ�2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g�����ʡ�H=2����-534kJ/��-67.7kJ/mol=-1135.7 kJ/mol��
�º�NO2����N2��ˮ�������Ȼ�ѧ����ʽΪ��2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g������H=-1135.7 kJ/mol��
A��ˮ��״̬ӦΪ��̬������Һ̬����A����
B����Ӧ��Ϊ��H=-1135.7 kJ/mol�����ǡ�H=-1000.3 kJ/mol����B����
C��1molN2H4��g����Ӧ��ˮ��״̬ӦΪ��̬������Һ̬����Ӧ��Ϊ��H=-567.86 kJ/mol����C����
D��������������֪���º�NO2����N2��ˮ�������Ȼ�ѧ����ʽΪ��2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g������H=-1135.7 kJ/mol����D��ȷ��
��ѡD��
���������⿼���Ȼ�ѧ����ʽ����д���ѶȲ���ע����д�������˹���ɵ����ã�

��ϰ��ϵ�д�
�����Ŀ



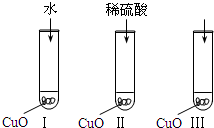 ��2007?��ͷ��ģ������ͭ��һ�ֺ�ɫ���壬������ϡ���ᣮijͬѧ��֪����ϡ������������ӣ�H2O��H+��SO42-����ʹ����ͭ�ܽ⣮�������һ��ͨ����ͼ��͢�����ʵ��������̽�����
��2007?��ͷ��ģ������ͭ��һ�ֺ�ɫ���壬������ϡ���ᣮijͬѧ��֪����ϡ������������ӣ�H2O��H+��SO42-����ʹ����ͭ�ܽ⣮�������һ��ͨ����ͼ��͢�����ʵ��������̽����� ��������ͪ�����ܾ��еĻ�ѧ������
��������ͪ�����ܾ��еĻ�ѧ������



