��Ŀ����
16����1����Ȼ��Cr��Ҫ��+3�ۺ�+6�۴��ڣ�+6�۵�Cr������ϸ����ͻ�䣬�������������ƽ��仹ԭΪ+3�۵ĸ�����ɲ���ƽ�������ӷ���ʽ��1Cr2O72-+3SO32-+8��=2Cr3++3SO42-+4H2O
��2��Ϊ�ⶨNiSO4•xH2O������x��ֵ����ȡ26.3g�����������ȫʧȥ�ᾧˮ��ʣ�����15.5g����ʽ����x��ֵ����6��
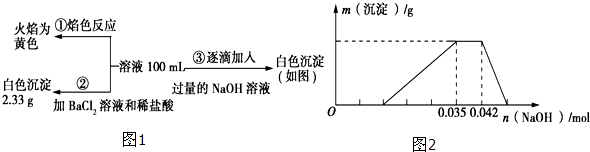
��3��ij��ɫ��ˮ�п��ܺ���H+��NH4+��Fe3+��Al3+��Mg2+��Na+��NO3-��CO32-��SO42-�еļ��֣�Ϊ������ɷ֣��ֱ�ȡ��ˮ��Ʒ100mL������������ʵ�飬��������й�ͼ����ͼ��ʾ��
��ش��������⣺
��ʵ����������1.0mol/L��NaOH��Һ100mL�������������˲�������������ƽ����Ͳ��ҩ�ס��ձ�����ͷ�ιܣ���ȱ�ٵ�����Ϊ100mL����ƿ
��ʵ����г�������A��B��������������Ӧ�����ӷ���ʽΪAl��OH��3+OH-=AlO2-+2H2O
�۸�������3��ʵ����Է�����ˮ��һ�������ڵ������У�Fe3+��Mg2+��CO32-��
�ܷ���ͼ����ԭ��Һ��c��NH4+����c��Al3+���ı�ֵΪ1��1��ȷ��NO3-�Ƿ���ڣ���ȷ��������ڡ����������ڡ���ȷ�������������ڣ��Լ���c��NO3-��=�����������ڣ����ʲ������𣩣�
���� ��1��������Ӧ�и�Ԫ�ػ��ϼ۱仯������������ԭ��Ӧ�е�ʧ�����غ���ɽ��ԭ�Ӹ����غ���д����ʽ��
��2��26.3g�����������ȫʧȥ�ᾧˮ��ʣ�����15.5g��ʧȥˮ������=26.3g-15.5g=10.8g��NiSO4������Ϊ15.5g���������ʵ����õ�1��x=$\frac{15.5g}{155g/mol}$��$\frac{10.8g}{18g/mol}$������õ�x��
��3����������һ�����ʵ���Ũ����Һ�IJ���ѡ����Ҫ��������
��ɫ��ˮȷ����Fe3+������ʵ���ȷ����Na+������ʵ���ȷ����SO42-������ʵ���ȷ����Al3+��һ������Fe3+��Mg2+����ΪCO32-��Al3+���ܹ��棬������CO32-������Һ�д��ڵ�����Ϊ��Al3+��NH4+��H+��SO42-���������ᱵ�������n��SO42-��������ͼ�����n��Al3+����n��H+�����ٸ��ݵ���غ�ȷ����û��NO3-��
��� �⣺��1����Ӧ�и�Ԫ�ػ��ϼ۴�+6�۽�Ϊ+3�ۣ���Ԫ�ش�+4�����ߵ�+6�ۣ����ݵ�ʧ�����غ㡢ԭ�Ӹ����غ㣬��Ӧ����ʽ��Cr2O72-+3SO32-+8H+=2Cr3++3SO42-+4H2O��
�ʴ�Ϊ��1��3��8H+��2��3��4��
��2��26.3g�����������ȫʧȥ�ᾧˮ��ʣ�����15.5g��ʧȥˮ������=26.3g-15.5g=10.8g��NiSO4������Ϊ15.5g���������ʵ����õ�1��x=$\frac{15.5g}{155g/mol}$��$\frac{10.8g}{18g/mol}$������õ�x=6��
�ʴ�Ϊ��6��
��3��������һ�����ʵ���Ũ����Һ�IJ��裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ��õ�����������������������ƽ����Ͳ��ҩ�ס��ձ�����ͷ�ιܡ�100����ƿ��
�ʴ�Ϊ��100mL����ƿ��
����������������������������Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��Al��OH��3+OH-=AlO2-+2H2O��
����ɫ��ˮȷ����Fe3+������ʵ���ȷ����Na+������ʵ���ȷ����SO42-������ʵ���ȷ����Al3+��һ������Fe3+��Mg2+����ΪCO32-��Al3+���ܹ��棬������CO32-������Һ�д��ڵ�����Ϊ��Na+��Al3+��NH4+��H+��SO42-����ˮ��һ�������ڵ�������Fe3+��Mg2+��CO32-��
�ʴ�Ϊ��Fe3+��Mg2+��CO32-��
��֪���ᱵ����Ϊ2.33g����n��SO42-��=$\frac{2.33g}{233g/mol}$=0.01mol��
����ͼ���֪��Al��OH��3��OH-Ϊ��n��OH-��=0.007mol��
Al��OH��3+OH-=AlO2-+2H2O
n��Al3+�� 0.007mol
����n��Al3+��=0.007mol���������ӳ�����Ҫ��������0.021mol��������Һ��H+������������0.014mol�������ӵ����ʵ�����0.014mol��
NH4++OH-=NH3•H2O��������������0.007mol������笠����ӵ����ʵ�����0.007mol��ԭ��Һ��c��NH4+����c��Al3+���ı�ֵΪ1��1��
���������ӵ�����ȷ�������Ը��ݵ���غ㣬��ȷ���Ƿ������������ӣ�
�ʴ�Ϊ��1��1����ȷ����
���� ����Ϊ�ۺ��⣬������������ԭ����ʽ��д�����ӷ�Ӧ���йؼ��㼰�жϡ�һ�����ʵ���Ũ����Һ�����ƣ���ȷ������ԭ��Ӧ��ʧ�����غ���ɣ���Ϥ�������ӵ����ʼ���Ӧ��ȷ����ͼ�����ǽ���ؼ�����Ŀ�Ѷ��еȣ�

| A�� | ���ܣ�H-F��H-Cl��H-Br��H-I | |
| B�� | ��ԭ�ԣ�Cl-��Br-��Fe2+��I- | |
| C�� | ��Ӧ�ȣ�C��s��+O2��g��=CO2��g����H1CO��g��+$\frac{1}{2}$O2��g��=CO2��g����H2��H1����H2 | |
| D�� | ���ԣ������������Һ��ͨ�������Ķ�����̼������ClO-+CO2+H2O=HCO${\;}_{3}^{-}$+HClO��˵�����ԣ�H2CO3��HClO��HCO${\;}_{3}^{-}$ |
| A�� | I-��I2 | B�� | Fe3+��Fe2+ | C�� | FeO��Fe | D�� | HCl��AgCl |
| A�� | CO2H2O | B�� | C2H4 CH4 | C�� | Cl2 C2H2 | D�� | NH3HCl |
| A�� | �⻯�� | B�� | ���۵⻯����Һ | C�� | ��� | D�� | ��ˮ |
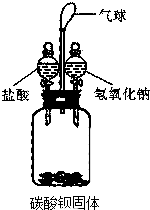 ij�о���ѧϰС������ͼ��ʾװ�ý�������ʵ�飨������������ˮ���εķ�Ӧ��
ij�о���ѧϰС������ͼ��ʾװ�ý�������ʵ�飨������������ˮ���εķ�Ӧ����1��������ƿ�м���һ����������Һ������ǡ��ȫ���ܽ�Ϊֹ���رջ��������۲쵽������Ȼ���ٽ�һ��������������Һ������ƿ�У��رջ������������������Ա�С����д�����������С�Ļ�ѧ��Ӧ����ʽ2NaOH+CO2=Na2CO3+H2O��ͬʱ�ڹ��ƿ�л��ܹ۲쵽�������Dz�����ɫ������
��2��ʵ�������ͬѧ�ǶԹ��ƿ�е���Һ����̽����
������⣺���ƿ�е���Һ������Щ���ʣ�
�������ۣ�������ҶԹ��ƿ���������ķ�Ӧ���з�������Ϊƿ��һ�����е�һ�������ǣ��ѧʽ����ͬ��NaCl��С��ȡ���ƿ����Һ�������Թ��У������м������ϡ���ᣬ���������ݲ������ɴ˵ó����ۣ����ƿ��Һ�л����е�������Na2CO3��С����Ϊ�����ܻ�����BaCl2�����һ����ΪС��Ľ����Ǵ���ģ�������BaCl2��Na2CO3����Һ�в����森������ۣ���ɹ�ʶ�����ƿ��Һ�л����ܴ���NaOH��
ʵ����ƣ�
���ǵ��Ѿ�ȷ����������������һ�����ʵĴ��ڶ��������Ƶļ�����ɸ��ţ���Ʒ����Ƚ�����ȥ���ټ��飮���岽�������
| ʵ��Ŀ�� | ʵ�鲽�� | ʵ������ | ʵ����� |
| ��ȥ������ | ��ȡ���ƿ����Һ�������Թ��У������м������CaCl2��Һ����ַ�Ӧ����� | �а�ɫ���� ���� | �����л�����NaOH |
| �����Ƿ� ����NaOH | ����Һ�еμӷ�̪��Һ | ��Һ���ɫ |
| A�� | Fe3+ | B�� | Fe2+ | C�� | Cu2+ | D�� | Zn2+ |