��Ŀ����
10�������������Ԫ�����ڱ���һ���֣���������Сд��ĸ�ֱ����һ�ֻ�ѧԪ�أ�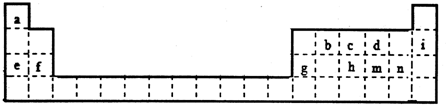
��1�������ϱ��ش��������⣺
�ٸ�����Ԫ�����γ���������������C����Ԫ�ط��ţ���m�������ӽṹʾ��ͼΪ
 ��
����h��m��n���γɵ�����������ˮ�����������ǿ������˳��ΪHClO4��H2SO4��H3PO4���ѧʽ��
��c��d��e��f��g�γɵļ����ӵİ뾶��С�����˳��ΪAl3+��Mg2+��Na+��O2-��N3-�������ӷ�����д����
��2�������ܹ������Ƚ�f��g���ʻ�����ǿ����ʵ�����BC��
A�����ڿ����з����Ѿõ�����Ԫ�صĿ�״���ʷֱ������ˮ��
B����������Ԫ�صĵ��ʷ�ĩ�ֱ��ͬŨ�ȵ����ᷴӦ
C����������Ԫ�صĵ��ʷ�ĩ�ֱ����ˮ���ã��������̪��Һ
D���Ƚ�������Ԫ�ص���̬�⻯����ȶ���
��3��a��d��e��m����Ԫ�ؿ���������п�ͼ�г�Br2��L��������ʣ�a��d���γɳ���Һ̬������K������A�к���eԪ�ص���һ�������ӣ���һ�������£����и����ʿɷ�������ͼ��ʾ�ı仯����Ӧ�����ɵ�ˮû��д������

����Ԫ�������ڱ��е�λ���ǵ������ڵڢ�A�壮
������A������ѧ������Ϊ���Ӽ����ۼ���
�۷�Ӧ�������ӷ���ʽΪSO2+Br2+2H2O=4H++SO42-+2Br-��
���� ����Ԫ�����ڱ���Ԫ�ص�λ�úͷֲ�������֪a��H��b��C��c��N��d��O��e��Na��f��Mg��g��Al��h��P��i��Ne��m��S��n��Cl��
��1����C�����γɶ����л�������SΪ16��Ԫ�أ��õ�2�������γ������8�����ӵ��ȶ��ṹ��
��Ԫ�صķǽ�����Խǿ��������������Ӧ��ˮ��������Խǿ��
�ۺ�������Ų���ͬ������ԭ������ԽС���뾶Խ��
��2��������Խǿ��ѧ����Խ���ã������ý�����ˮ���ᷴӦ������֤��
��3��a��d��e��m�ֱ�ΪH��O��Na��S��H��O���γɳ���Һ̬������K��H����K�пɵ���Һ����KΪH2O������A�к��б�Ԫ�ص���һ�������ӣ�AΪNa2O2����BΪNaOH��CΪO2��FΪSO2���ݴ˽�ɣ�
��� �⣺����Ԫ�����ڱ���Ԫ�ص�λ�úͷֲ�������֪a��H��b��C��c��N��d��O��e��Na��f��Mg��g��Al��h��P��i��Ne��m��S��n��Cl��
��1��������ЩԪ���У�C���γɶ����л���������������SΪ16��Ԫ�أ��õ�2�������γ������8�����ӵ��ȶ��ṹ�����ӽṹʾ��ͼΪ�� ��
��
�ʴ�Ϊ��C�� ��
��
������Cl��S����P����ͬһ���ڣ�Ԫ�صķǽ�����Cl��S��P��������������Ӧˮ��������ԣ�HClO4��H2SO4��H3PO4���ʴ�Ϊ��HClO4��H2SO4��H3PO4��
�����������γɵĺ�������Ų���ͬ������ԭ������Al��Mg��Na��O��N�������Ӱ뾶��Al3+��Mg2+��Na+��O2-��N3-���ʴ�Ϊ��Al3+��Mg2+��Na+��O2-��N3-��
��2��������Mg��Al����Mg�Ļ�ѧ���ʱ�Al���ã������ý�����ˮ�����ᣩ��Ӧ�����׳̶ȡ����ҳ̶Ƚ�����֤����ѡBC��
��3��a��d��e��m�ֱ�ΪH��O��Na��S��H��O���γɳ���Һ̬������K��H����K�пɵ���Һ����KΪH2O������A�к��б�Ԫ�ص���һ�������ӣ�AΪNa2O2����BΪNaOH��CΪO2��FΪSO2
���崦�ڵ������ڵڢ�A�壬�ʴ�Ϊ���������ڵڢ�A�壻
��AΪ�������ƣ��������ƺ������Ӽ����ۼ����ʴ�Ϊ�����Ӽ����ۼ���
�۶����������巴Ӧ������������ᣬ���ӷ�ӦΪSO2+Br2+2H2O=4H++SO42-+2Br-���ʴ�Ϊ��SO2+Br2+2H2O=4H++SO42-+2Br-��
���� ���⿼��ѧ��Ԫ�����ڱ���Ԫ�������ɵ��ۺ�֪ʶ�����Ը�����ѧ֪ʶ���лش��ѶȲ���

 ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д� A�ӽ��� ϵ�д�
A�ӽ��� ϵ�д� ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д�| A�� | 0.1mol/L����15mL | B�� | 0.2mol/L����12mL | ||
| C�� | 0.15mol/L����8mL | D�� | 18mol/L����15mL |
| A�� | 9.6g | B�� | 9.8g | C�� | 19.2g | D�� | ��ȷ�� |
| Ԫ�ر�� | Ԫ�����ʻ�ԭ�ӽṹ |
| T | M���ϵ����������ڲ��������3�� |
| X | �����������Ǵ�����������2�� |
| Y | �����µ���Ϊ˫ԭ�ӷ��ӣ����⻯��ˮ��Һ�ʼ��� |
| Z | Ԫ�����������+7�� |
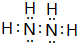 ��
����2����Ԫ�ط��ű�ʾ��������Ԫ��ԭ�Ӱ뾶����Դ�СS��Cl��C��N��
��3��Ԫ��Z��Ԫ��T��ȣ��ǽ����Խ�ǿ����Cl����Ԫ�ط��ű�ʾ�������б�������֤����һ��ʵ����ab
a��һ��������Z�ĵ��ʺ�T�ĵ��ʷֱ���Fe������Ӧ�������Ͳ�ͬ
b��T���⻯��ˮ��Һ�ڿ����з������ױ��ʣ���Z���⻯��ˮ��Һ���ױ���
c��Ԫ��Z��T������������Ӧˮ���ﶼ��������������Һ��Ӧ
��4��Ԫ��T���⻯����������������Ӧ��ˮ�����Ũ��Һ���������·�Ӧ��������һ�������д���÷�Ӧ�Ļ�ѧ����ʽH2S+3H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$4SO2+4H2O��
| A�� | ��ϵͳ������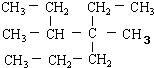 ������Ϊ4��5-����-4-�һ����� ������Ϊ4��5-����-4-�һ����� | |
| B�� | 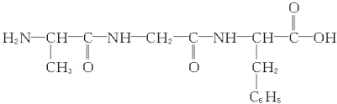 ��ȫˮ����Եõ�3�ְ����� ��ȫˮ����Եõ�3�ְ����� | |
| C�� | ʯ���ѽ����֬���������ɸ߷�����������С�������ʵĹ��� | |
| D�� | ������ ��һ�ȴ�����2�� ��һ�ȴ�����2�� |
| A�� | Ϊ��ֹ����е��ؽ�������Ⱦ������ˮ�壬Ӧ���������ϵ�ص��ۺ����ü��� | |
| B�� | �ߴ��ȵĶ�������㷺�����������ά����ǿ��ᡰ��·�� | |
| C�� | ��NO��N02Ϊ���ĵ����������γɹ⻯ѧ�����������һ����Ҫԭ�� | |
| D�� | FeCl3��Һ����Cu��Ӧ��������ʴ��ӡˢ��· |
| A�� | ��ϩ���ӵ����ʽ��CH2 | B�� | ����ķ���ʽ��C6H12 | ||
| C�� | �ǻ��Ļ�ѧʽ��OH- | D�� | 1��2-��������Ľṹ��ʽ��C2H4Br2 |