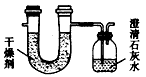��Ŀ����
��֪ij��ȼ�Ϻ���̼���⡢��3��Ԫ�ء�Ϊ�˲ⶨ����ȼ����̼��������Ԫ�ص������ȣ��ɽ���̬ȼ�Ϸ���������������ȼ�գ���ʹ����������ȫ��ͨ����ͼ��ʾ��װ�ã��õ����±����е�ʵ����(���������������ȫ������)

|
|
ʵ��ǰ |
ʵ��� |
|
(�������U�ι�)������ |
101.1 g |
102.9 g |
|
(ʯ��ˮ�����ƿ)������ |
312.0 g |
314.2 g |
����ʵ��������
(1)ʵ����Ϻ���������ˮ����Ϊ________g��������ƿ������һ�����Σ�������Ϊ________g��
(2)���ɵ�ˮ����Ԫ�ص�����Ϊ________g��
(3)���ɵĶ�����̼��̼Ԫ�ص�����Ϊ________g��
(4)��ȼ����̼Ԫ������Ԫ�ص�������Ϊ________��
(5)��֪����ȼ�ϵ�ÿ�������к���һ����ԭ�ӣ����ȼ�ϵķ���ʽΪ________���ṹ��ʽΪ________��
(1)1.8��5��(2)0.2 ��(3)0.6��(4)3��1 (5)CH4O��CH3OH
��������(1)m(H2O)��102.9 g��101.1 g��1.8 g
m(CO2)��314.2 g��312.0 g��2.2 g
��n(CaCO3)��n(CO2)��0.05 mol
m(CaCO3)��5 g
(2)m(H)��m(H2O)��2/18��1.8 g��2/18��0.2 g
(3)m(C)��m(CO2)��12/44��2.2 g��12/44��0.6 g
(4)m(C)��m(H)��0.6 g��0.2 g��3��1
(5)��ȼ�Ϸ�����C��H��ԭ�Ӹ�����Ϊ��n(C)��n(H)��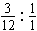 ��1��4
��1��4
��̼�ļ۵�ԭ��֪�����л�������е�̼��ԭ�Ӹ�����Ϊ1��4ʱ��������ֻ�ܺ�CH4��������ΪCH4��������������Ϊÿ�������к���һ����ԭ�ӣ����ȼ�ϵķ���ʽΪCH4O���ṹ��ʽΪCH3OH��

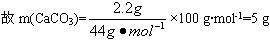
| ʵ��ǰ | ʵ��� |
(�����+U�ι�)������ | ||
(ʯ��ˮ+���ƿ)������ |
����ʵ����������������⣺
(1)ʵ����Ϻ���������ˮ������Ϊ_______________g��������ƿ������һ�����Σ�������Ϊ_______________ g��
(2)���ɵ�ˮ����Ԫ�ص�����Ϊ_______________ g��
(3)���ɵĶ�����̼��̼Ԫ�ص�����Ϊ_______________ g��
(4)��ȼ����̼Ԫ������Ԫ�ص�������Ϊ_______________��
(5)��֪����ȼ�ϵ�ÿ��������ֻ�ܺ���һ����ԭ�ӣ����ȼ�ϵķ���ʽΪ______________,�ṹ��ʽΪ_______________��
��֪ij��ȼ�Ϻ���̼���⡢������Ԫ�ء�Ϊ�˲ⶨ����ȼ����̼��������Ԫ�ص������ȣ��ɽ���̬ȼ�Ϸ���������������ȼ�գ���ʹ����������ȫ��ͨ������ͼ��ʾ��װ�ã��õ����±����е�ʵ���������������������ȫ�����գ���
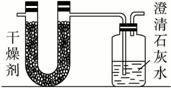
| ʵ��ǰ | ʵ��� | |
| �������+U�ιܣ������� | 101.1 g | 102.9 g |
| ��ʯ��ˮ+���ƿ�������� | 312.0 g | 314.2 g |
����ʵ����������������⣺
��1��ʵ����Ϻ���������ˮ������Ϊ_______________g��������ƿ������һ�����Σ�������Ϊ_______________ g��
��2�����ɵ�ˮ����Ԫ�ص�����Ϊ_______________ g��
��3�����ɵĶ�����̼��̼Ԫ�ص�����Ϊ_______________ g��
��4����ȼ����̼Ԫ������Ԫ�ص�������Ϊ_______________��
��5����֪����ȼ�ϵ�ÿ��������ֻ�ܺ���һ����ԭ�ӣ����ȼ�ϵķ���ʽΪ______________,�ṹ��ʽΪ_______________��
| ʵ��ǰ | ʵ��� |
�������+U�ιܣ������� | 101.1 g | 102.9 g |
��ʯ��ˮ+���ƿ�������� | 312.0 g | 314.2 g |
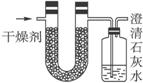
����ʵ��������
��1��ʵ����Ϻ���������ˮ������Ϊ_________g��������ƿ������һ�����Σ�������Ϊ_________g��
��2����ȼ����̼Ԫ������Ԫ�ص�������Ϊ_________��
��3����֪����ȼ�ϵ�ÿ�������к���һ����ԭ�ӣ����ȼ�ϵķ���ʽΪ_________���ṹ��ʽΪ_________��