��Ŀ����
ij��ѧ��ȤС��ⶨijFeCl3��Ʒ��������FeCl2���ʣ�����Ԫ�ص�����������ʵ���Ұ����²�����У�
�ٳ���ag��Ʒ�������ձ��У�
�ڼ��������������������ˮ��ʹ��Ʒ�ܽ⣬Ȼ��ȷ���Ƴ�250 mL��Һ��
��ȷ��ȡ25.00 mL���������õ���Һ�������ձ� �У�������������ˮʹ��Ӧ��ȫ��
�ܳ���Ѹ�ټ��������ˮ����ֽ��裬ʹ������ȫ��
�ݹ��ˣ�ϴ�ӳ�����
������ת�Ƶ������ڣ����ȡ����裬ֱ������ȫ���ɺ��ɫ��Ϊ����ɫ�����ڸ���������ȴ�����º�����
���ظ����ϲ�������������γƵõ����������0.1 gΪֹ��
����������������ش�
(1)��ͼ��ʾ�����У���ʵ�鲽��٢ڢ��б����õ���������E��F��____������ĸ����
�ٳ���ag��Ʒ�������ձ��У�
�ڼ��������������������ˮ��ʹ��Ʒ�ܽ⣬Ȼ��ȷ���Ƴ�250 mL��Һ��
��ȷ��ȡ25.00 mL���������õ���Һ�������ձ� �У�������������ˮʹ��Ӧ��ȫ��
�ܳ���Ѹ�ټ��������ˮ����ֽ��裬ʹ������ȫ��
�ݹ��ˣ�ϴ�ӳ�����
������ת�Ƶ������ڣ����ȡ����裬ֱ������ȫ���ɺ��ɫ��Ϊ����ɫ�����ڸ���������ȴ�����º�����
���ظ����ϲ�������������γƵõ����������0.1 gΪֹ��
����������������ش�
(1)��ͼ��ʾ�����У���ʵ�鲽��٢ڢ��б����õ���������E��F��____������ĸ����

(2)����ڼ��������Ŀ����_____________���ӿ���Ʒ�ܽ���������_____________________��
(3)д��������з�����Ӧ�����ӷ���ʽ___________________��
(4)����ݵ�ϴ����ϴȥ�����ڳ����ϵ�________��д���ӷ��ţ���ϴ�ӳ����IJ���Ϊ__________��
(5)������������W1����������Ⱥ�������������W2������Ʒ����Ԫ�ص���������Ϊ________��
(3)д��������з�����Ӧ�����ӷ���ʽ___________________��
(4)����ݵ�ϴ����ϴȥ�����ڳ����ϵ�________��д���ӷ��ţ���ϴ�ӳ����IJ���Ϊ__________��
(5)������������W1����������Ⱥ�������������W2������Ʒ����Ԫ�ص���������Ϊ________��
(1)C��G
(2)��ֹFeCl3��FeCl2ˮ�⣻�ò���������
(3)2Fe2++Cl2=2Fe3++2Cl-
(4)NH4+��Cl-��OH-����©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2~3��
(2)��ֹFeCl3��FeCl2ˮ�⣻�ò���������
(3)2Fe2++Cl2=2Fe3++2Cl-
(4)NH4+��Cl-��OH-����©����ע������ˮ��ʹˮ��û����������ˮ��Ȼ������ظ�����2~3��
(5)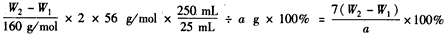
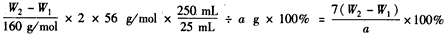

��ϰ��ϵ�д�
�����Ŀ




