��Ŀ����
14����ú��Ϊȼ�Ͽ�ͨ����������;����;����C��s��+O2��g���TCO2��g������H1��0��
;�������Ƴ�ˮú����
C��s��+H2O��g���TCO��g��+H2��g������H2��0 ��
��ȼ��ˮú����
2CO��g��+O2��g���T2CO2��g������H3��0��
2H2��g��+O2��g���T2H2O��g������H4��0 ����ش��������⣺
��1��;����ų������������ϵ��ڣ�����ڡ������ڡ���С�ڡ���;����ų���������
��2����H1����H2����H3����H4����ѧ��ϵʽ�ǡ�H1=��H2+$\frac{1}{2}$����H3+��H4����
��3����֪����C��s��+O2��g��=CO2��g������H=-393.5kJ•mol?1
��2CO��g��+O2��g��=2CO2��g������H=-566kJ•mol?1
��TiO2��s��+2Cl2��g��=TiCl4��s��+O2��g������H=+141kJ•mol?1
��TiO2��s��+2Cl2��g��+2C��s��=TiCl4��s��+2CO��g���ġ�H=-80kJ•mol-1��
��4����֪���и����Ȼ�ѧ����ʽ
��1��Fe2O3��s��+3CO��g��=2Fe��s��+3CO2��H=-25kJ/mol
��2��3Fe2O3��s��+CO��g��=2Fe3O4��s��+CO2��g����H=-47kJ/mol
��3��Fe3O4��s��+CO��g��=3FeO��s��+CO2��g����H=+640kJ/mol
��д��FeO��s����CO��g����ԭ��Fe��CO2��g�����Ȼ�ѧ����ʽCO��g��+FeO��s��=Fe��s��+CO2��g����H=-1308kJ/mol��
���� ��1�����ݸ�˹���ɵ�ԭ�����жϣ�
��2�����ݸ�˹�������жϸ�����Ӧ֮��Ĺ�ϵ��
��3�����ݸ�˹���ɣ�����֪�Ȼ�ѧ����ʽ�����ʵ���ϵ�����мӼ�����Ӧ��Ҳ������Ӧ��ϵ��������Ӧ�ļӼ���
��4������д��FeO��s����CO��ԭ��Fe��CO2���Ȼ�ѧ����ʽCO��g��+FeO��s��=Fe��g��+CO2��g����H�����ø�˹���ɢ١�3-���ۡ�2+�ڣ��ɵõ��û�ѧ����ʽ���Դ˼����H��
��� �⣺��1���ɸ�˹���ɿ�֪������һ����Ӧ���Էֲ����У��������Ӧ�����ջ�ų��������ܺ��������Ӧһ�η���ʱ���ջ�ų���������ͬ��
�ʴ�Ϊ�����ڣ�
��2�����ݸ�˹���ɣ���Ӧ1=��Ӧ2+��Ӧ3��$\frac{1}{2}$+��Ӧ4��$\frac{1}{2}$���ԡ�H1=��H2+$\frac{1}{2}$����H3+��H4����
�ʴ�Ϊ����H1=��H2+$\frac{1}{2}$����H3+��H4����
��3����֪����C��s��+O2��g��=CO2��g������H=-393.5kJ•mol-1
��2CO��g��+O2��g��=2CO2��g������H=-566kJ•mol-1
��TiO2��s��+2Cl2��g��=TiCl4��s��+O2��g������H=141kJ•mol-1
���ݸ�˹���ɣ��١�2-��+�۵�TiO2��s��+2Cl2��g��+2C��s��=TiCl4��s��+2CO��g����
�ʡ�H=2����-393.5kJ•mol-1-��-566kJ•mol-1��+141kJ•mol-1=-80kJ•mol-1��
�ʴ�Ϊ��-80kJ•mol-1��
��4����Fe2O3��s��+3CO��g��=2Fe��s��+3CO2��g������H=-25kJ/mol
��3Fe2O3��s��+CO��g��=2Fe3O4��s��+CO2��g������H=-47kJ/mol
��Fe3O4��s��+CO��g��=3FeO��s��+CO2��g������H=+640kJ/mol
���ݸ�˹���ɢ١�3-���ۡ�2+�ڣ��õ���
6CO��g��+6FeO��s��=6Fe��g��+6CO2��g����H=-66kJ/mol��
�õ��Ȼ�ѧ����ʽΪ��CO��g��+FeO��s��=Fe��s��+CO2��g����H=-1308kJ/mol��
�ʴ�Ϊ��CO��g��+FeO��s��=Fe��s��+CO2��g����H=-1308kJ/mol��
���� ���⿼���˹���ɡ���Ӧ�ȵ��йؼ��㡢�Ȼ�ѧ����ʽ����д���Ѷ��еȣ�ע���˹���ɵ��������Ȼ�ѧ����ʽ����д��

| A�� | ��ˮ������Ҵ�������ȡ�������� | |
| B�� | ������ε���Ũ�����У����÷�ˮԡ������ȡ������ | |
| C�� | ��ͭ˿�ھƾ����ϼ��Ⱥ�����������ˮ�Ҵ��У�ͭ˿�ָ�ԭ���ĺ�ɫ | |
| D�� | ���������ˮ������Һ��������������Һ��ˮԡ���ȣ��ɿ������������� |
| A�� | C6H12O6��������aq��+6O2$\stackrel{ø}{��}$6CO2+6H2O | |
| B�� | CH3COOH+KOH�TCH3COOK+H2O | |
| C�� | A+B�TC+D����H��0 | |
| D�� | Ba��OH��2•8H2O��NH4Cl��Ӧ |
| A�� | ���ǿ��������Ƚ��Ļ�ѧ�������豸�����µ�ԭ�� | |
| B�� | ���ǿ������ô���ʹˮ������� | |
| C�� | ���ǿ��������Ƚ��ļ������豸�����µķ��� | |
| D�� | ��ѧ��ѧֻ��ͨ��ʵ����̽�����ʵ����� |
��SiO2 ��H2SiO3 ��Fe��OH��3 ��CuS ��FeCl2 ��CaSiO3��
| A�� | �ڢ� | B�� | �ڢܢ� | C�� | �ڢۢܢ� | D�� | �ڢܢ� |
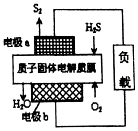
| A�� | �缫aΪ��صĸ��� | |
| B�� | �缫b�Ϸ����ĵ缫��ӦΪO2+4H++4e-�T2H2O | |
| C�� | ����·��ͨ��2mol���ӣ������ڲ��ͷ�632kJ���� | |
| D�� | ����17gH2S���뷴Ӧ�������1molH+������Ĥ���������� |

��֪��25��ʱ����������������ܶȻ�������Ksp�������ʾ��
| �������� | Al��OH��3 | Fe��OH��3 | Cu��OH��2 | Mn��OH��2 |
| Ksp | 1.0��10-33 | 4.0��10-38 | 2.0��10-20 | 4.0��10-14 |
��1������1�Ļ�ѧʽΪCaSO4��
��2����NH4��2S�ĵ���ʽΪ
 ����������ʱ�����루NH4��2S������ΪʹCu2+ת��ΪCuS������
����������ʱ�����루NH4��2S������ΪʹCu2+ת��ΪCuS��������3�����ữ����ԭ���У�����������������ԭ��Ӧ�����ӷ���ʽΪ��
��4����֪����Һ3�г�MnSO4�⣬������������NH4��2SO4����NH4��2SO4��MnSO4���ܽ��������ͼ2��ʾ��
�ݴ��жϣ�������I��ӦΪ����Ũ�������ȹ��ˡ�ϴ�ӡ����
��5����ҵ�Ͽ��õ������MnSO4��Һ�ķ����Ʊ�MnO2����������ӦʽΪMn2++2H2O-2e-=MnO2+4H+��
��6��25.35g MnSO4•H2O��Ʒ���ȷֽ���̵��������ߣ���Ʒ�������¶ȱ仯�����ߣ���ͼ3��ʾ��
��300��ʱ�����ù���Ļ�ѧʽΪMnSO4��
��1150��ʱ����Ӧ�Ļ�ѧ����ʽΪ3MnO2$\frac{\underline{\;1150��\;}}{\;}$Mn3O4+O2����
