��Ŀ����
ˮ�ĵ���ƽ��������ͼ��ʾ��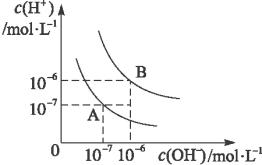
(1)����A���ʾ25 ��ʱˮ�ĵ���ƽ��ʱ������Ũ�ȣ����¶����ߵ�100 ��ʱ��ˮ�ĵ���ƽ��״̬��B�㣬���ʱˮ�����ӻ���_______________���ӵ�____________��
(2)��pH=8��Ba(OH)2��Һ��pH=5��ϡ�����ϣ�������100 ��ĺ��£���ʹ���Һ��pH=7����Ba(OH)2����������֮��Ϊ______________��
(3)��֪AnBm�����ӻ�=��C(Am+)��n����C(Bn-)��m��ʽ�е�C(Am+)��C(Bn-)��ʾ���ӵ����ʵ���Ũ�ȡ���ij�¶���Ca(OH)2�ܽ��Ϊ0.74 g���䱥����Һ�ܶ���Ϊ1 g��mL-1�������ӻ�ԼΪ________________��
(1)1��10-14 1��10-12 (2)2��9 (3)0.004
������100 ��ʱ��pH=6��������Һ��(2)�еĻ����ҺpH=7�ʼ��ԣ�
��c(OH-)=10-7 mol��L-1
=![]() ��
��
�������ɵó���������ȡ�
(3)��Ca(OH)2�����ӻ�=c(Ca2+)����c(OH-)��2��
c��Ca(OH)2��=![]() ��0.1 mol��L-1��
��0.1 mol��L-1��
��c(Ca2+)=0.1 mol��L-1��c(OH-)=0.2 mol��L-1��
������ʽ���ɽ��Ca(OH)2�����ӻ���

��ϰ��ϵ�д�
�����Ŀ
 ˮ�ĵ���ƽ��������ͼ��ʾ��
ˮ�ĵ���ƽ��������ͼ��ʾ�� ��2006?�ɶ�ģ�⣩ˮ�ĵ���ƽ��������ͼ��ʾ��
��2006?�ɶ�ģ�⣩ˮ�ĵ���ƽ��������ͼ��ʾ�� ��2012?����һģ����1��ˮ�ĵ���ƽ��������ͼ��ʾ����A���ʾ25��ʱˮ�ĵ����ƽ��ʱ������Ũ�ȣ�B���ʾ100��Cʱˮ�ĵ����ƽ��ʱ������Ũ�ȣ�100��ʱ1mol?L-1 ��NaOH��Һ�У���ˮ�������c��H+��=
��2012?����һģ����1��ˮ�ĵ���ƽ��������ͼ��ʾ����A���ʾ25��ʱˮ�ĵ����ƽ��ʱ������Ũ�ȣ�B���ʾ100��Cʱˮ�ĵ����ƽ��ʱ������Ũ�ȣ�100��ʱ1mol?L-1 ��NaOH��Һ�У���ˮ�������c��H+��= ��2011?��̨ģ�⣩ˮ�ĵ���ƽ��������ͼ��ʾ��
��2011?��̨ģ�⣩ˮ�ĵ���ƽ��������ͼ��ʾ��