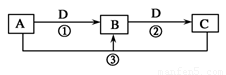
��Ŀ����
��֪A��B��C��Ϊ�����ĵ��ʣ�����AΪ������B��CΪ�ǽ�������һ���������ת����ϵ��ͼ��ʾ����Ӧ�����Ͳ��ֲ�����ʡ�ԣ���
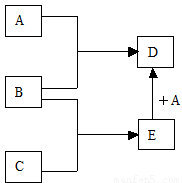
��ش�
��1����������B��C��Ϊ���壬DΪ��ɫ���壬E�Ļ�ѧʽΪ______��A��E��һ�������·�Ӧ����D�Ļ�ѧ����ʽΪ_____________��
��2����������BΪ���壬CΪ��ɫ���壬�ɽ���A��ԭ�ӽṹʾ��ͼΪ_______��E�Ļ�ѧʽΪ___________��A��E��һ�������·�Ӧ����D�Ļ�ѧ����ʽΪ___________��
��1��H2O 3Fe��4H2O������ Fe3O4��4H2
Fe3O4��4H2
��2��
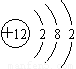 ��CO2�� 2Mg��CO2
��CO2�� 2Mg��CO2 2MgO��C
2MgO��C
��������
�����������֪A��B��C��Ϊ�����ĵ��ʣ�����AΪ������B��CΪ�ǽ�������һ���������ת������������B��C��Ϊ���壬DΪ��ɫ���壬�������뵽����ˮ�����ķ�Ӧ������E�Ļ�ѧʽΪH2O��A��E��һ�������·�Ӧ����D�Ļ�ѧ����ʽΪ3Fe��4H2O������ Fe3O4��4H2 ����������BΪ���壬CΪ��ɫ���壬�������뵽þ�Ͷ�����̼�ķ�Ӧ�����Թ��ɽ���A��ԭ�ӽṹʾ��ͼΪ
Fe3O4��4H2 ����������BΪ���壬CΪ��ɫ���壬�������뵽þ�Ͷ�����̼�ķ�Ӧ�����Թ��ɽ���A��ԭ�ӽṹʾ��ͼΪ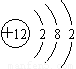 ��E�Ļ�ѧʽΪCO2��A��E��һ�������·�Ӧ����D�Ļ�ѧ����ʽΪ2Mg��CO2
��E�Ļ�ѧʽΪCO2��A��E��һ�������·�Ӧ����D�Ļ�ѧ����ʽΪ2Mg��CO2 2MgO��C��
2MgO��C��
���㣺����ת�����ƶ�
������һ����ԣ��ƶ����˼ά�����ɷ����֣�һ��˳��˼ά������������һ�����Ƴ�δ֪��Ϣ���ڶ���������˼ά�����������������ƣ������裻���������Ǵ��Լ��ҵ���ͻ�ƿڽ��з�ɢ�Ƶ������ƶ���ʱ����������ͬʱ�ҵ�����ͻ�ƿڣ��Ӽ��������������֣�����ƶϡ�����˵��ѧ�ƶ���û�нݾ���̸������Ҫ������ѵ�����ܽᾭ�顢Ѱ�ҹ��ɣ����ֲ�����ٻع�α����ٽ���ѵ����������������˶�Ϊ�����ƶ������С��о�����

 ��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�
��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�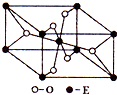 ��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ��ǰ�����ڣ�Aλ�����ڱ���s������ԭ���е��Ӳ�����δ�ɶԵ�������ͬ��B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3����A��B��D����Ԫ����ɵ�һ�ֻ�����M����װ�����г����е�һ���к����壬A��B����Ԫ����ɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����N�dz������л��ܼ���E�С����������֮�ƣ�E4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��
��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ��ǰ�����ڣ�Aλ�����ڱ���s������ԭ���е��Ӳ�����δ�ɶԵ�������ͬ��B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3����A��B��D����Ԫ����ɵ�һ�ֻ�����M����װ�����г����е�һ���к����壬A��B����Ԫ����ɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����N�dz������л��ܼ���E�С����������֮�ƣ�E4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��