��Ŀ����
����Ŀ��[��ѧ����ѡ��3�����ʽṹ������]�⻯���ƣ�NaAlH4����һ���������ʴ�����ϣ���������Ti��NaAlH4��150��ʱ���⣬��170�桢15.2MPa���������ظ����⡣NaAlH4����AlCl3��NaH���ʵ������ºϳɡ�NaAlH4�ľ����ṹ������ͼ��ʾ��

��1����̬Tiԭ�ӵļ۵��ӹ����ʾʽΪ ��
��2��NaH���۵�Ϊ800�棬�������л��ܼ���NaH���� ������������ʽΪ ��
��3��AlCl3��178��ʱ����������������Է�������ԼΪ267���������ӵĽṹʽΪ ��������λ������
��4��AlH4���У�Al�Ĺ���ӻ���ʽΪ ��������AlH4���ռ乹����ͬ���������� ������ѧʽ����
��5��NaAlH4����������Na+�����ҵȾ��AlH4���� ����NaAlH4������ܶ�Ϊ g��cm��3���ú�a�Ĵ���ʽ��ʾ������NaAlH4�������Ĵ���Na+��Li+ȡ�����õ��ľ���Ϊ ������ѧʽ����
��6��NaAlH4���������Ϊ��ÿ3��AlH4��������2���ֱ��ͷ���3��Hԭ����1��Alԭ����ͬʱ���Alԭ������ڵ�Naԭ��ת�������ͷŵ�Alԭ�����µĿ�λ���γ��µĽṹ�����ֽṹ�仯�ɱ������չ���������壬�����ͷų����������������������ѧ����ʽ��ʾΪ ��
���𰸡���1��![]()
��2��������![]()
��3�� 
��4��sp3 NH4+��BH4������SO42����PO43�� �����������𰸣�
��5��8 ������
������ ![]() ���� Na3Li(AlH4)4
���� Na3Li(AlH4)4
��6��3NaAlH4��Na3AlH6+2Al+3H2��
��������
�����������1��Ti��ԭ������Ϊ22�������Ų�Ϊ1s22s22p63s23p63d24s2���۵����Ų�ʽΪ3d24s2���۵��ӹ����ʾʽΪ![]() ��
��
��2��NaH���۵�Ϊ800�����������л��ܼ����������Ӿ��壻NaH�����ӻ��������ʽΪ![]() ��
��
��3���Ȼ�������ԭ����������ֻ��3�����ӣ��γ�3�����ۼ���ÿ����ԭ�Ӻ��ĸ���ԭ���γɹ��ۼ���������һ�����õ��Ӷ�����ԭ���ṩ�γɵ���λ������ͼ ��
��
��4��AlH4-�У�Al�ļ۲���Ӷ���=4+![]() =4������Alԭ��Ϊsp3�ӻ����ȵ�����Ľṹ���ƣ�����AlH4-�ռ乹����ͬ����������NH4+��BH4-������SO42-����PO43-������
=4������Alԭ��Ϊsp3�ӻ����ȵ�����Ľṹ���ƣ�����AlH4-�ռ乹����ͬ����������NH4+��BH4-������SO42-����PO43-������
��5���Ե���Na+�����о�����֮�����AlH4-����Ϊ![]() anm��λ��ͬһ���4�������Լ�����4������������ϣ�����8�������ݾ�̯����֪�������������ӵĸ���Ϊ6��
anm��λ��ͬһ���4�������Լ�����4������������ϣ�����8�������ݾ�̯����֪�������������ӵĸ���Ϊ6��![]() +4��
+4��![]() =4������Ļ�ѧʽΪNaAlH4�����Ծ�����AlH4-�ĸ���Ϊ4���þ���������Ϊ
=4������Ļ�ѧʽΪNaAlH4�����Ծ�����AlH4-�ĸ���Ϊ4���þ���������Ϊ![]() g���þ������Ϊ2a3nm3=2a3��10-21cm3����þ������ܶ�Ϊ
g���þ������Ϊ2a3nm3=2a3��10-21cm3����þ������ܶ�Ϊ ��������AlH4-�ĸ���Ϊ4�������ӵĸ���Ϊ4����NaAlH4�������Ĵ���Na+��Li+ȡ��������AlH4-�ĸ���Ϊ4�������ӵĸ���Ϊ3������ӵĸ���Ϊ1������Ļ�ѧʽΪ��Na3Li(AlH4)4��
��������AlH4-�ĸ���Ϊ4�������ӵĸ���Ϊ4����NaAlH4�������Ĵ���Na+��Li+ȡ��������AlH4-�ĸ���Ϊ4�������ӵĸ���Ϊ3������ӵĸ���Ϊ1������Ļ�ѧʽΪ��Na3Li(AlH4)4��
��6��NaAlH4���������Ϊ��ÿ3��AlH4-�У���2���ֱ��ͷų�3��Hԭ�Ӻ�1��Alԭ�ӣ�ͬʱ���Alԭ������ڵ�Naԭ��ת�Ƶ����ͷŵ�Alԭ�����µĿ�λ����������Al��H2��AlH63-������ʽΪ3NaAlH4��Na3AlH6+2Al+3H2����

 �������ϵ�д�
�������ϵ�д�����Ŀ���±�ΪԪ�����ڱ���һ���֣������Ԫ�آ٣����ڱ��е�λ�ã��ش����⣺
�� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
1 | �� | |||||||
2 | �� | �� | �� | |||||
3 | �� | �� | �� | �� | �� |
��1���������ڰ뵼����ϵ�Ԫ�������ڱ��е�λ����__________________��
��2���ۡ��ܡ����ԭ�Ӱ뾶��С��___________________����Ԫ�ط����ش𣩡�
��3���ݡ��ޡ��ߵ�����������Ӧ��ˮ���������ǿ����__________������ѧʽ�ش𣩡�
��4���ڡ��ۡ��ܵ���̬�⻯��ȶ�����ǿ����__________�����ṹʽ�ش�
��5���ں͢۰�ԭ����1:2�γɵĻ����������ʽΪ____________���þ��������Ĺ����п�
��������������Ϊ_______________________��
��6���ۺ͢��γɵĻ���������_______________������ӻ�������ۻ���������þ�������________���壨����ӡ��������ӡ�����ԭ�ӡ�����
��7��Ԫ�آݡ��ߵ�����������ˮ���ﻥ�෴Ӧ�Ļ�ѧ����ʽΪ��___________________��
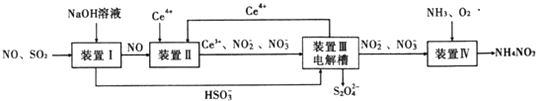
����Ŀ�������ҹ�����ӵ�����ʽϿ��������ƣ�����β���ѳ�Ϊ��Ҫ�Ŀ�����Ⱦ��ش��������⣺
��l����������������ʱ������Ӧ��N2(g)��O2(g)![]() 2NO(g)���ǵ�������β���к���NO��ԭ��֮һ��2000Kʱ�����ݻ�Ϊ2L���ܱ������г���2molN2��2molO2������������Ӧ������5min�ﵽƽ�⣬��ʱ������NO���������Ϊ0.75% ����÷�Ӧ��5min�ڵ�ƽ����Ӧ����v(O2)=_______mol/(Lmin)��N2��ƽ��ת����Ϊ_______��2000Kʱ�÷�Ӧ��ƽ�ⳣ��K=_____��
2NO(g)���ǵ�������β���к���NO��ԭ��֮һ��2000Kʱ�����ݻ�Ϊ2L���ܱ������г���2molN2��2molO2������������Ӧ������5min�ﵽƽ�⣬��ʱ������NO���������Ϊ0.75% ����÷�Ӧ��5min�ڵ�ƽ����Ӧ����v(O2)=_______mol/(Lmin)��N2��ƽ��ת����Ϊ_______��2000Kʱ�÷�Ӧ��ƽ�ⳣ��K=_____��
��2��һ����NO�����ֽ�Ĺ����У�NO��ת������ʱ��仯�Ĺ�ϵ����ͼ��ʾ��

�� ��Ӧ2NO(g)![]() N2(g)��O2(g)Ϊ______��Ӧ������ȡ����ȡ�) ��
N2(g)��O2(g)Ϊ______��Ӧ������ȡ����ȡ�) ��
��һ���¶��£��ܹ�˵����Ӧ2NO(g)![]() N2(g)��O2(g)�Ѵﵽƽ�����_____������ţ���
N2(g)��O2(g)�Ѵﵽƽ�����_____������ţ���
a.�����ڵ�ѹǿ�������仯
b�����������ܶȲ������仯
c. 2NO��N2��O2��Ũ�ȱ��ֲ���
d����λʱ���ڷֽ�4molNO��ͬʱ����2molN2
�� ���ĸ��ݻ����¶Ⱦ���ȫ��ͬ���ܱ������зֱ�����������ʣ���Ӧ���ʵ�����mol�����±���ʾ����ͬ�����´ﵽƽ���N2���������������_______�����������ţ���
�������� | NO | N2 | O2 |
A | 2 | 0 | 0 |
B | 0 | l | l |
C | 0.4 | 0.6 | 0.8 |
D | l | 0.5 | 0.4 |
��3��������������ϡ��ȼ��ʱ��β���е���Ҫ��Ⱦ��ΪNOx������CH4����ԭNOx����������������Ⱦ��
��֪��CH4(g)+4NO2(g)=4NO(g)+CO2(g)+2H2O(g)��H=-574kJ��mol-1
CH4(g)+2NO2(g)=N2(g)+CO2(g)+2H2O(g)��H=-867kJ��mol-1
��д��CH4��NO��Ӧ����N2��CO2��H2O(g)���Ȼ�ѧ����ʽ��___________��
��ʹ�ô������Խ�����β������Ҫ�к��ɷ�һ����̼��CO���͵������NOx��ת��Ϊ�����壬�÷�Ӧ�Ļ�ѧ����ʽΪ___________________��