��Ŀ����
16����ϩ����ϩ����ʯ���ѽ�IJ���֮һ���ش��������⣺��1����ϩ��ʹ��ˮ��ɫ��д���䷴Ӧ�Ļ�ѧ����ʽCH2=CH2+Br2��CH2BrCH2Br��
��2����һ�������£���ϩ�������Ȼ��⡢�����ȷ�����Ӧ�⣬��ϩ���Ӽ仹���Է����Ӿ۷�Ӧ����д
���䷴Ӧ�Ļ�ѧ����ʽnCH2=CHCH3$\stackrel{һ������}{��}$
 ����ϩ�����Ӿ۷�Ӧ�õ��IJ��ﲻ�ܣ�����ܡ������ܡ���ʹ����KMnO4��Һ��ɫ����ϩ�������ܹ���ƽ���ԭ�����Ϊ7 ����
����ϩ�����Ӿ۷�Ӧ�õ��IJ��ﲻ�ܣ�����ܡ������ܡ���ʹ����KMnO4��Һ��ɫ����ϩ�������ܹ���ƽ���ԭ�����Ϊ7 ����
���� ��1����ϩ���Ժ���ˮ�����ӳɷ�Ӧ������1��2-�������飻
��2����ϩ�к���̼̼˫�������Է����Ӿ۷�Ӧ���۱�ϩ�в�����̼̼˫����������̼̼˫�������ʣ�������ϩ�Ĺ���ṹ�жϱ�ϩ��ԭ�ӹ��������
��� �⣺��1����ϩ���Ժ���ˮ�����ӳɷ�Ӧ��ʹ��ˮ��ɫ������1��2-�������飬��ӦΪCH2=CH2+Br2��CH2BrCH2Br���ʴ�Ϊ��CH2=CH2+Br2��CH2BrCH2Br��
��2����ϩ����ϩ��Ϊͬϵ���ϩ�����Ӿ۷�Ӧ�ķ���ʽ�ǣ�nCH2=CHCH3$\stackrel{һ������}{��}$ ���Ӿ۲����в�����̼̼˫�������Ա�ϩ�����Ӿ۷�Ӧ�õ��IJ��ﲻ��ʹ����KMnO4��Һ��ɫ����ϩ�������ܹ���ƽ���ԭ�����Ϊ7����������ϩ�ṹ��ͬ��3��̼ԭ���Լ�3����ԭ�ӣ����м��ϵ�һ����ԭ�ӣ��ʴ�Ϊ��nCH2=CHCH3$\stackrel{һ������}{��}$
���Ӿ۲����в�����̼̼˫�������Ա�ϩ�����Ӿ۷�Ӧ�õ��IJ��ﲻ��ʹ����KMnO4��Һ��ɫ����ϩ�������ܹ���ƽ���ԭ�����Ϊ7����������ϩ�ṹ��ͬ��3��̼ԭ���Լ�3����ԭ�ӣ����м��ϵ�һ����ԭ�ӣ��ʴ�Ϊ��nCH2=CHCH3$\stackrel{һ������}{��}$ �����ܣ�7��
�����ܣ�7��
���� ���⿼���л���������Լ�ԭ�ӹ�����жϺ��л�����ʽ����д���ѶȲ���ע�����֪ʶ���������գ�

��ϰ��ϵ�д�
 �����ҵ���������ͯ������ϵ�д�
�����ҵ���������ͯ������ϵ�д�
�����Ŀ
6�������й�ʵ��������ʹ�÷�����ʵ�������ȷ���ǣ�������
| A�� | �����ᾧˮ�ⶨʵ���У�������ǯȡ��������������������ȴ | |
| B�� | ���Թܼмг��Թܼ���ʱ�����ֽ�����ס�Թܼеij����Ͷ̱� | |
| C�� | �ⶨ�кͷ�Ӧ�ķ�Ӧ��ʱ����NaOH��Һһ���Ե��������У����û��β��������������Һ | |
| D�� | ʵ���ҴӺ�������ȡ���ʵ�ķ����ǣ�ȡ�������ա��ܽ�����ˡ���ȡ������ |
7����֪NH4CuSO3��������10mol/L������Һ����ȣ��������������к�ɫ�������ɢڲ����̼�����ζ������ ����Һ����ɫ���ݴ��ж�����˵����ȷ���ǣ�������
| A�� | ��Ӧ�������������� | |
| B�� | NH4CuSO3����Ԫ�ر����� | |
| C�� | �̼�����ζ�������ǰ��� | |
| D�� | 1 mol NH4CuSO3��ȫ��Ӧת��0.5 mol���� |
4�������йص������Һ��˵����ȷ���ǣ�������
| A�� | ������ˮ�еμ�ŨH2SO4��KW���� | |
| B�� | ����AgCl��AgI���������Һ��c��Ag+����c��Cl-��=c��I-�� | |
| C�� | pH��ͬ�Ģ�CH3COONa��NaHCO3��NaClO������Һ��c��Na+�����٣��ڣ��� | |
| D�� | �����£�pH=2��������pH=12�İ�ˮ�������ϣ�������Һ�У�c��Cl-����c��NH4+����c��H+����c��OH-�� |
11������˵����ȷ���ǣ�������
| A�� | �ᡢ��ζ��ǵ���� | |
| B�� | H2O��D2O����ͬ�������� | |
| C�� | ʯ�͵ķ�����ѻ������ڻ�ѧ�仯 | |
| D�� | ֻ�����ۼ������ʣ�һ���ǹ��ۻ����� |
1������˵����ȷ���ǣ�������
| A�� | ��ϵͳ��������������  ������Ϊ2��6-����-5-�һ����� ������Ϊ2��6-����-5-�һ����� | |
| B�� | ���Ϸ���ʽΪC3H4Cl2������ͬ���칹����ĿΪ7�� | |
| C�� | ���顢�����顢�����顢����ķе��������� | |
| D�� | 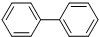 �� �� �ֱ���������Ͷ������飬��Ϊͬϵ����ַ��ӵ�����ԭ�Ӷ��п��ܹ�ƽ�� �ֱ���������Ͷ������飬��Ϊͬϵ����ַ��ӵ�����ԭ�Ӷ��п��ܹ�ƽ�� |
8��25��ʱ������˵��һ����ȷ���ǣ�������
| A�� | ��pH=4����Һ�У�ˮ����������ܵ�c��OH-����10-10 mol•L-1 | |
| B�� | ��pH=2������Һ��pH=12�ļ���Һ�������Ϻ���Һ��pH=7 | |
| C�� | ��pH=4�������pH=4�Ĵ���ֱ�������NaHCO3��Ӧ�����߲���CO2�� | |
| D�� | ��pH=4�����pH=10�������������������ϣ�c��Na+��+c��H+��=c��OH-��+c��CH3COO-�� |
5����1.01��105Pa��120��ʱ��1���ij����4���������ϣ���ȫȼ�պ�ָ���ԭ���¶Ⱥ�ѹǿ��������䣬����������̼ԭ�����������ǣ�������
| A�� | 1 | B�� | 2 | C�� | 3 | D�� | 4 |
20���������𱽷ӡ��Ҵ����������ơ������������軯��������Һ��һ���Լ��ǣ�������
| A�� | ��ˮ | B�� | ����Cu��OH��2 | C�� | FeCl3��Һ | D�� | ������ |