��Ŀ����
�����������������ʸֲĵ�����������ij���ߵ���Ҫ�ɷ�Ϊ����M��Ca��������3. 5%������������
CaO��
(1)CaԪ�������ڱ���λ����___________��
(2)Ca������õķǽ���Ԫ��A�γɻ�����D��D�ĵ���ʽΪ______________��D�ķе��A��Si�γɵĻ�����E�ķе�___________��
(3)��ƽ�ø����������Ļ�ѧ����ʽ
___P+ ___FeO+___CaO ___Ca3(PO4)2+___Fe
___Ca3(PO4)2+___Fe
(4)��������������ϡ����������NaOH��Һ�����ɰ�ɫ��״������Ѹ�ٱ�ɻ���ɫ������ɺ��ɫM(OH)n������Mn+���Լ���______________��
(5)ȡ1.6 g������������ˮ��ַ�Ӧ������224 mL H2����״������������Һ��ͨ��������CO2������ܵõ�CaCO3____g��
CaO��
(1)CaԪ�������ڱ���λ����___________��
(2)Ca������õķǽ���Ԫ��A�γɻ�����D��D�ĵ���ʽΪ______________��D�ķе��A��Si�γɵĻ�����E�ķе�___________��
(3)��ƽ�ø����������Ļ�ѧ����ʽ
___P+ ___FeO+___CaO
 ___Ca3(PO4)2+___Fe
___Ca3(PO4)2+___Fe (4)��������������ϡ����������NaOH��Һ�����ɰ�ɫ��״������Ѹ�ٱ�ɻ���ɫ������ɺ��ɫM(OH)n������Mn+���Լ���______________��
(5)ȡ1.6 g������������ˮ��ַ�Ӧ������224 mL H2����״������������Һ��ͨ��������CO2������ܵõ�CaCO3____g��
(1)�������ڢ�A��
(2)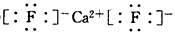 ����
����
(3)2P+5FeO+3CaO 1Ca3(PO4)2+5Fe
1Ca3(PO4)2+5Fe
(4)KSCN(��NaOH������������)
(5)1.1
(2)
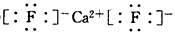 ����
����(3)2P+5FeO+3CaO
 1Ca3(PO4)2+5Fe
1Ca3(PO4)2+5Fe(4)KSCN(��NaOH������������)
(5)1.1

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ




 Ԫ�������ڱ���λ���� ����ԭ�ӽṹʾ��ͼ ��
Ԫ�������ڱ���λ���� ����ԭ�ӽṹʾ��ͼ ��
