��Ŀ����
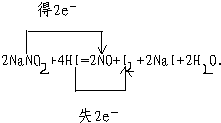
���������ƣ�NaNO2���е�Ԫ�صĻ��ϼ�Ϊ+3�ۣ�Ϊ֤�����о��������Ƶ����ʣ��������м����Լ��ɹ�ѡ��
��KMnO4��Һ����ϡH2SO4��Һ����ϡHNO3��Һ����ϡNaOH��Һ���ݵ��ۡ�KI��Һ����Ʒ����Һ����ע��NaNO2�Ļ�ԭ�����е�Ԫ�صĻ��ϼ�һ��Ϊ+2�ۣ�
���������Լ��ش����и��ʣ�
��1��֤��NaNO2����������Ӧѡ����Լ���______��������Ӧ�����ӷ���ʽ��________��
��2��֤��NaNO2���л�ԭ��Ӧѡ����Լ���______��������Ӧ�����ӷ���ʽ��______��
��1���ڢݡ� 2NO2�� +2I��+4H+==I2+2NO��+2H2O
��2���٢� 5NO2��+2MnO4��+6H+==5NO3��+2Mn2++3H2O

��ϰ��ϵ�д�
 ��ѧ�����ϵ�д�
��ѧ�����ϵ�д� �·Ƿ��̸����100��ϵ�д�
�·Ƿ��̸����100��ϵ�д�
�����Ŀ
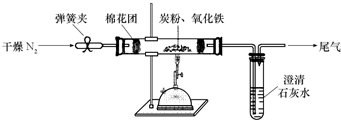

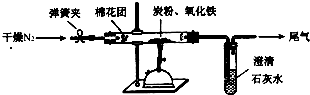 ij�о���ѧϰС��Թ���̿������������Ӧ���������ɷֽ����о���
ij�о���ѧϰС��Թ���̿������������Ӧ���������ɷֽ����о���