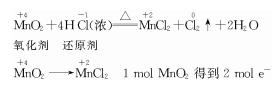��Ŀ����
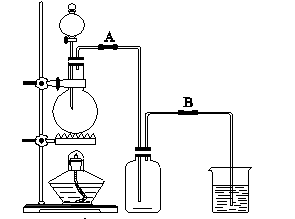
ijУ��ѧ��ȤС����Ũ������������̼��ȷ�Ӧ����ȡ���ռ�������װ�����£�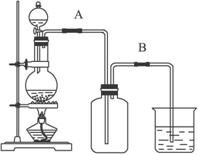
(1)д���÷�Ӧ�Ļ�ѧ����ʽ��____________________________________��
(2)������Ӧ�У���ԭ����_________������1 mol�������μӷ�Ӧ��ת�Ƶĵ��ӵ����ʵ�����_________mol��
(3)ʵ�鿪ʼǰ��ijѧ����װ�ý����������Լ�顣�����ǣ����Ӻ�װ��֮���õ��ɼм�ס_________��(�A����B��)��Ƥ�ܣ����Һ©���м�ˮ�����������ӵ�һ����ʱ��ֹͣ��ˮ�����ù۲졣���۲쵽��Һ©����_________ʱ��������װ�ò�©����
(4)��Ҫ�ռ�����������������������ռ�֮ǰ��������ͨ����ϴ��ƿ����ϴ��ƿ��ʢ�Լ�������__________________��_________________��
(5)��ȤС��ͬѧ�����ձ��м���������Һ�е�һ�������ղ�������������Ϊ���д������__________(��д���)��
A.����������Һ B.̼������Һ C.Ũ����
(1)4HCl(Ũ)+MnO2![]() MnCl2+2H2O+Cl2��
MnCl2+2H2O+Cl2��
(2)HCl(�����Ũ����)��2 mol
(3)B Һ�治�½�
(4)ʳ��ˮ Ũ����
(5)C
���������⿼��Cl2��ʵ�����Ʒ�,4HCl(Ũ)+MnO2![]() MnCl2+2H2O +Cl2��,�ɷ�Ӧԭ����֪,��������MnO2,��ԭ����HCl,1 mol�������μӷ�Ӧ��ת�Ƶĵ��ӵ����ʵ�����2 mol��
MnCl2+2H2O +Cl2��,�ɷ�Ӧԭ����֪,��������MnO2,��ԭ����HCl,1 mol�������μӷ�Ӧ��ת�Ƶĵ��ӵ����ʵ�����2 mol��
�������������Ʊ������ǰ��,Ӧ��סB������Ƥ�ܣ����������ԽϺã����Է�Һ©����Һ�治�½����ռ�����֮ǰӦ���ñ���ʳ��ˮ����(HCl)������Ũ��������ˮ��������������Һ���ն����Cl2��ã�̼������ҺҲ���ԣ�Ũ����С�

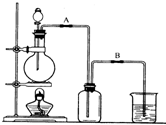 ijУ��ѧ��ȤС����Ũ������������̼��ȷ�Ӧ����ȡ���ռ�������װ�����£�
ijУ��ѧ��ȤС����Ũ������������̼��ȷ�Ӧ����ȡ���ռ�������װ�����£�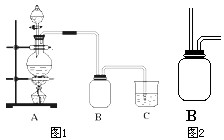 ijУ��ѧ��ȤС����Ũ������������̼��ȷ�Ӧ����ȡ���ռ�������װ����ͼ1��ʾ��
ijУ��ѧ��ȤС����Ũ������������̼��ȷ�Ӧ����ȡ���ռ�������װ����ͼ1��ʾ��