��Ŀ����
�����£���16mol N2��b mol H2�Ļ������ͨ��һ�������ݻ����ܱ������У��������·�Ӧ��N2��g��+3H2��g��?2NH3��g��
��1������Ӧ���е�ijʱ��tʱ��nt��H2��=15mol��nt��NH3��=6mol����b��ֵΪ
��2����Ӧ�ﵽƽ��ʱ�������������Ϊ716.8L������£�������NH3�ĺ��������������Ϊ25%������ƽ��ʱ��
��ԭ���������ƽ��������������ʵ���֮�ȣ�д����������ȣ���ͬ��
�ڴﵽƽ��ʱ��N2��H2��ת����֮��a��N2����a��H2��
��ƽ���������У�n��N2����n��H2����n��NH3��
��1������Ӧ���е�ijʱ��tʱ��nt��H2��=15mol��nt��NH3��=6mol����b��ֵΪ
24
24
����2����Ӧ�ﵽƽ��ʱ�������������Ϊ716.8L������£�������NH3�ĺ��������������Ϊ25%������ƽ��ʱ��
��ԭ���������ƽ��������������ʵ���֮�ȣ�д����������ȣ���ͬ��
�ڴﵽƽ��ʱ��N2��H2��ת����֮��a��N2����a��H2��
��ƽ���������У�n��N2����n��H2����n��NH3��
��������1���������ɰ��������ʵ�������������ʽ���㣻
��2���������������ܵ����ʵ�������������������㰱�������ʵ�����
�ٸ��ݷ�Ӧ����ƽ��ʱ�ܵ����ʵ����������ò���������ԭ�����������ʵ�����
�ڸ��ݲ������������Ӧ���ĵĵ��������������ʵ����������ת���ʣ�
�۸��ݢ٢������ݼ�����ﵽƽ��ʱ����Ӧ��������ֵ����ʵ������ݴ˼��㣮
��2���������������ܵ����ʵ�������������������㰱�������ʵ�����
�ٸ��ݷ�Ӧ����ƽ��ʱ�ܵ����ʵ����������ò���������ԭ�����������ʵ�����
�ڸ��ݲ������������Ӧ���ĵĵ��������������ʵ����������ת���ʣ�
�۸��ݢ٢������ݼ�����ﵽƽ��ʱ����Ӧ��������ֵ����ʵ������ݴ˼��㣮
����⣺��1�����ݷ���ʽ���㣺
N2��g��+3H2��g��?2NH3��g��
��ʼ��mol����16mol b 0
ת����mol����3mol 9mol 6mol
tʱ�̣�mol����15mol 6mol
����b-9=15����b=24��
�ʴ�Ϊ��24��
��2����Ӧ��ƽ��ʱ���������Ϊ
=32mol������NH3�����ʵ���Ϊ32mol��25%=8mol��
�����ò��������㣺
N2 ��g��+3H2��g��?2NH3��g�������ʵ������١�n
1mol 3mol 2mol 2mol
ת����4mol 12mol 8mol 8mol
��ԭ�������Ϊ��32mol+8mol=40mol��
��ԭ���������ƽ������������ʵ���֮��n��ʼ����n��ƽ��=40mol��32mol=5��4��
��ԭ���������ƽ��������������ʵ���֮��Ϊ5��4��
�ڷ�Ӧת���ĵ������ʵ���Ϊ4mol��ת����Ϊ��
��100%=25%��
ת�����������ʵ���Ϊ12mol��ת����Ϊ��
��100%=50%��
�ﵽƽ��ʱ��N2��H2��ת����֮��a��N2����a��H2��=25%��50%=1��2��
�𣺴ﵽƽ��ʱ��N2��H2��ת����֮��a��N2����a��H2��Ϊ1��2��
��ƽ�����������Ϊ��N2Ϊ16mol-4mol=12 mol��NH3Ϊ8 mol��H2Ϊ32mol-12mol-8mol=12mol��
ƽ���������У�n��N2����n��H2����n��NH3��=12mol��12mol��8mol=3��3��2��
��ƽ���������У�n��N2����n��H2����n��NH3��Ϊ3��3��2��
N2��g��+3H2��g��?2NH3��g��
��ʼ��mol����16mol b 0
ת����mol����3mol 9mol 6mol
tʱ�̣�mol����15mol 6mol
����b-9=15����b=24��
�ʴ�Ϊ��24��
��2����Ӧ��ƽ��ʱ���������Ϊ
| 716.8L |
| 22.4L/mol |
�����ò��������㣺
N2 ��g��+3H2��g��?2NH3��g�������ʵ������١�n
1mol 3mol 2mol 2mol
ת����4mol 12mol 8mol 8mol
��ԭ�������Ϊ��32mol+8mol=40mol��
��ԭ���������ƽ������������ʵ���֮��n��ʼ����n��ƽ��=40mol��32mol=5��4��
��ԭ���������ƽ��������������ʵ���֮��Ϊ5��4��
�ڷ�Ӧת���ĵ������ʵ���Ϊ4mol��ת����Ϊ��
| 4mol |
| 16mol |
ת�����������ʵ���Ϊ12mol��ת����Ϊ��
| 12mol |
| 24mol |
�ﵽƽ��ʱ��N2��H2��ת����֮��a��N2����a��H2��=25%��50%=1��2��
�𣺴ﵽƽ��ʱ��N2��H2��ת����֮��a��N2����a��H2��Ϊ1��2��
��ƽ�����������Ϊ��N2Ϊ16mol-4mol=12 mol��NH3Ϊ8 mol��H2Ϊ32mol-12mol-8mol=12mol��
ƽ���������У�n��N2����n��H2����n��NH3��=12mol��12mol��8mol=3��3��2��
��ƽ���������У�n��N2����n��H2����n��NH3��Ϊ3��3��2��
���������⿼�黯ѧƽ��ļ��㣬��Ŀ�ѶȲ���ע����������ʽ����ϻ�ѧ����ʽ���㣮

��ϰ��ϵ�д�
 Ӯ�ڿ�����ʦ��ʱ�ƻ�ϵ�д�
Ӯ�ڿ�����ʦ��ʱ�ƻ�ϵ�д� �������Ͽ�ʱͬ��ѵ��ϵ�д�
�������Ͽ�ʱͬ��ѵ��ϵ�д�
�����Ŀ

 �����ϵ��SO3�İٷֺ������¶ȵĹ�ϵ����ͼ��ʾ���������κ�һ�㶼��ʾƽ��״̬��������ͼʾ�ش��������⣺
�����ϵ��SO3�İٷֺ������¶ȵĹ�ϵ����ͼ��ʾ���������κ�һ�㶼��ʾƽ��״̬��������ͼʾ�ش��������⣺

 �ġ�H��������0���>��
�ġ�H��������0���>�� 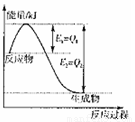

 �����ϵ��SO3�İٷֺ������¶ȵĹ�ϵ����ͼ��ʾ���������κ�һ�㶼��ʾƽ��״̬��������ͼʾ�ش��������⣺
�����ϵ��SO3�İٷֺ������¶ȵĹ�ϵ����ͼ��ʾ���������κ�һ�㶼��ʾƽ��״̬��������ͼʾ�ش��������⣺ 
 �ġ�H��������0���>��
�ġ�H��������0���>�� 
 ��ʱ��KSP(AgCl)=1.6��10-10mol2��L-2��KSP��AgI��=1.5��10-16mol��L-2����25���£���100mL 0.002mol��L-1��NaCl��Һ����μ���100mL 0.002mol��L-1��������Һ���а�ɫ�������ɡ��ӳ����ܽ�ƽ��ĽǶȽ��Ͳ���������ԭ����������������������������Ӧ�����Һ�У���������0.1mol��L��1��NaI��Һ�������������������������������������������������ԭ���ǣ������ӷ���ʽ��ʾ������������������������������������������������������ ��
��ʱ��KSP(AgCl)=1.6��10-10mol2��L-2��KSP��AgI��=1.5��10-16mol��L-2����25���£���100mL 0.002mol��L-1��NaCl��Һ����μ���100mL 0.002mol��L-1��������Һ���а�ɫ�������ɡ��ӳ����ܽ�ƽ��ĽǶȽ��Ͳ���������ԭ����������������������������Ӧ�����Һ�У���������0.1mol��L��1��NaI��Һ�������������������������������������������������ԭ���ǣ������ӷ���ʽ��ʾ������������������������������������������������������ ��