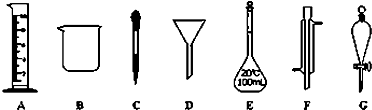
��Ŀ����
ʵ��������100mL 0.1mol?L-1 ��Na2CO3��Һ�����ȡ________��Na2CO3?10H2O���壬��ȷʵ�����˳����________��
A������������ˮϴ���ձ��Ͳ�����2-3�Σ�ϴ��Һת��������ƿ��
B������Ʒ�����ձ���������ˮ�ܽ⣬��ȴ�����º�ת��������ƿ��
C��ѡ��100mL����ƿ���������Ƿ�©ˮ
D���ý�ͷ�ι�������ƿ�м�ˮ���̶���
E��������ƿ�м�ˮ����̶���1-2cm��
F��������ƿ���ã�����ҡ�ȣ�
2.9 CBAEDF
����������n=cV��������100mL 0.1mol?L-1 ��Na2CO3��Һ��ҪNa2CO3�����ʵ�����̼�������ʵ�������̼���ƾ�������ʵ������ٸ���m=nM������Ҫ̼���ƾ����������
����ʵ��ʵ�ʲ�������ʵ�����˳����������Ʋ����м��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����
�������100mL 0.1mol?L-1 ��Na2CO3��Һ��ҪNa2CO3�����ʵ���Ϊ��0.1L��0.1mol?L-1=0.01mol��Na2CO3?10H2O�����ʵ�������Na2CO3�����ʵ���������Na2CO3?10H2O������0.01mol��286g/mol=2.9g��
���Ʋ����м��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣬���ò��������裬��ȴ��ת�Ƶ�100mL����ƿ�У����ò���������������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμ����̶��ߣ����Ǻ�ƿ��ҡ�ȣ���ʵ�����˳��Ϊ��CBAEDF��
�ʴ�Ϊ��2.9��CBAEDF��
���������⿼����һ�����ʵ���Ũ����Һ�����ƣ��ѶȲ���ע��������ʵ�����ʱҪ�������ʺ�Na2CO3.10H2O�Ĺ�ϵ��
����������n=cV��������100mL 0.1mol?L-1 ��Na2CO3��Һ��ҪNa2CO3�����ʵ�����̼�������ʵ�������̼���ƾ�������ʵ������ٸ���m=nM������Ҫ̼���ƾ����������
����ʵ��ʵ�ʲ�������ʵ�����˳����������Ʋ����м��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����
�������100mL 0.1mol?L-1 ��Na2CO3��Һ��ҪNa2CO3�����ʵ���Ϊ��0.1L��0.1mol?L-1=0.01mol��Na2CO3?10H2O�����ʵ�������Na2CO3�����ʵ���������Na2CO3?10H2O������0.01mol��286g/mol=2.9g��
���Ʋ����м��㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ�����ձ����ܽ⣬���ò��������裬��ȴ��ת�Ƶ�100mL����ƿ�У����ò���������������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμ����̶��ߣ����Ǻ�ƿ��ҡ�ȣ���ʵ�����˳��Ϊ��CBAEDF��
�ʴ�Ϊ��2.9��CBAEDF��
���������⿼����һ�����ʵ���Ũ����Һ�����ƣ��ѶȲ���ע��������ʵ�����ʱҪ�������ʺ�Na2CO3.10H2O�Ĺ�ϵ��

��ϰ��ϵ�д�
 �п�������㾫��ϵ�д�
�п�������㾫��ϵ�д�
�����Ŀ
ʵ��������100mL 1.0mol/L Na2CO3��Һ�������й�������ȷ���ǣ�������
| A����������ƽ��ȡ10.6g Na2CO3���� | B����Na2CO3�����������ƿ�У���ˮ���̶��� | C������ʱ����Һ�棬������ҺŨ��ƫ�� | D����ˮʱ���������̶��ߣ��ý�ͷ�ι�ֱ����������Һ�� |