��Ŀ����
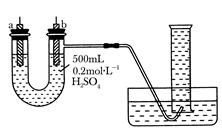
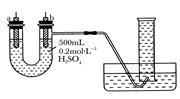
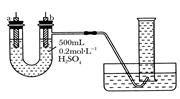
�����ṩ��п����ͭƬ��500 mL 0.2 mol/L��H2SO4��Һ�����ߡ�1 000 mL��Ͳ��������ͼװ�����ⶨп��ϡ���ᷴӦʱ����ij��ʱ����ͨ�����ߵĵ��ӵ����ʵ�����
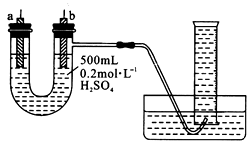
(1)����ͼ��ʾ��װ�����������ã���1 000mL��Ͳ���ѳ�����ˮ����ʼʵ��ʱ������Ҫ
__________________��
(2)��a�缫����Ϊ______����缫��ӦʽΪ_____________��
��b�缫����Ϊ______����缫��ӦʽΪ_______________��
(3)����Ͳ���ռ���672 mL����ʱ�������㵽��״���£���ͨ�����ߵĵ��ӵ����ʵ���Ϊ
__________��
__________________��
(2)��a�缫����Ϊ______����缫��ӦʽΪ_____________��
��b�缫����Ϊ______����缫��ӦʽΪ_______________��
(3)����Ͳ���ռ���672 mL����ʱ�������㵽��״���£���ͨ�����ߵĵ��ӵ����ʵ���Ϊ
__________��
(1)�õ��߰�a��b���缫��������
(2)��пƬ��Zn-2e-=Zn2+����ͭƬ��2H++2e-=H2��
(3)0.06 mol
(2)��пƬ��Zn-2e-=Zn2+����ͭƬ��2H++2e-=H2��
(3)0.06 mol

��ϰ��ϵ�д�
�����Ŀ