��Ŀ����
��֪��2CO(g)+O2(g)=2CO2(g)��H=-566 kJ/mol; Na2O2(s)+CO(g)=Na2CO3(s)��H=-226 kJ/mol ���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ����
A.CO��ȼ����Ϊ283 kJ
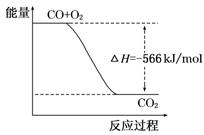
B.��ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������ϵ
C.2Na2O2(s)+2CO(s)=2Na2CO3(s) ��H��-452 kJ/mol
D.CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ 6.02��1023
C
����:ȼ��������һ��������1mol��ȼ����ȫȼ�������ȶ���������ʱ���ų����������䵥λ��kJ/mol��A����ȷ��B��û��ע����Ӧ������ʵ�����Ӧ����2molCO��1mol��������2mol������̼ʱ�ķ�Ӧ���̺�������ϵ������ȷ�������������Ȼ�ѧ����ʽ�ɵõ�2Na2O2(s)+2CO(g)=2Na2CO3(s) ��H��-452 kJ/mol�����������ڹ�̬ʱ������Ҫ��������̬ʱ�������������ڷ�Ӧ2Na2O2(s)+2CO(s)=2Na2CO3(s)�зų�������Ҫ�٣�����H��-452 kJ/mol���ڷ�ӦNa2O2(s)+CO(s)=Na2CO3(s)��ÿ����1molCO�����ת��2mol���ӣ���ת��1mol���ӣ�������0.5molCO���ų���ȼ����113kJ������C��

 ��ѧȫ��������ѵ��ϵ�д�
��ѧȫ��������ѵ��ϵ�д�