��Ŀ����
ij�л���A��C��H��O��N����Ԫ����ɣ����������ֱ�ΪC��54.8%,H:5.58%��N:7.11%������ΪO��A����Է�������������300��A�ǰ����ᣬ���ҷ����С�NH2�͡�COOH����ͬһ��̼ԭ���ϣ������в����ڼ�����CH3��������FeCl3��Һ������ɫ��Ӧ��1 mol A����Һ����3 mol NaOH��ȫ��Ӧ��������������⣺
��1���л���A����Է�������Ϊ________________�������ʽΪ_________________��
��2�����������������л���A�Ľṹ���֣�д�����е����ֵĽṹ��ʽ��________________��
________________��________________��
��1��197C9H11NO4
��2��6 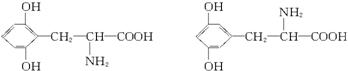
 �������������𰸣�
�������������𰸣�
����:
A�����е�C��H��O��N��ԭ�Ӹ�����Ϊ��n(C)��n(H)��n(O)��n(N)= ![]() =9��11��4��1��
=9��11��4��1��
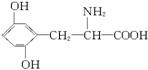
��֪A����Է�������������300����A����2��Nԭ�ӣ�����![]() ��300������A����һ��Nԭ�ӣ����Է���ʽΪC9H11NO4����Է�������Ϊ196��A�������С�NH2����COOH����OH��1 mol A����3 mol NaOH���ã�������������OH����A�Ľṹ��ʽΪ��
��300������A����һ��Nԭ�ӣ����Է���ʽΪC9H11NO4����Է�������Ϊ196��A�������С�NH2����COOH����OH��1 mol A����3 mol NaOH���ã�������������OH����A�Ľṹ��ʽΪ��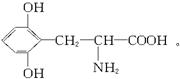

��ϰ��ϵ�д�
�����Ŀ
