��Ŀ����
14�����������ƣ�Na2S2O5���dz��õ�ʳƷ��������֮һ��ij�о�С���������ʵ�飺��ʵ��һ�����������Ƶ���ȡ
������ͼ1װ�ã�ʵ��ǰ�ѳ���װ���ڵĿ�������ȡNa2S2O5��װ�â�����Na2S2O5���������������ķ�ӦΪNa2SO3+SO2�TNa2S2O5��
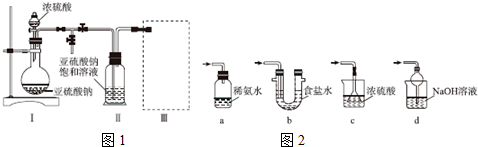
��1��װ�â��в�������Ļ�ѧ����ʽΪNa2SO3+H2SO4=Na2SO4+SO2��+H2O��
��2��Ҫ��װ�â��л���������ľ��壬�ɲ�ȡ�ķ��뷽���ǹ��ˣ�
��3��װ�â����ڴ���β������ѡ�õ������װ�ã��г���������ȥ��Ϊd������ţ���
��ʵ��������������Ƶ����ʣ�Na2S2O5����ˮ������NaHSO3��
��4��֤��NaHSO3��Һ��HSO${\;}_{3}^{-}$�ĵ���̶ȴ���ˮ��̶ȣ��ɲ��õ�ʵ�鷽����ae������ţ���
a���ⶨ��Һ��pH����b������Ba��OH��2��Һ
c���������� d������Ʒ����Һ
e������ɫʯ����ֽ���
��5�����Na2S2O5�����ڿ������ѱ�������ʵ�鷽����ȡ����Na2S2O5�������Թ��У���������ˮ�ܽ⣬�μ����ᣬ���ٵμ��Ȼ�����Һ���а�ɫ��������
��ʵ���������Ѿ��п��������������IJⶨ
��6�����ѾƳ���Na2S2O5�������������ⶨij���Ѿ��п��������IJ�������������SO2���㣩�ķ������£�

����֪���ζ�ʱ��Ӧ�Ļ�ѧ����ʽΪSO2+I2+2H2O�TH2SO4+2HI��
�ٰ���������ʵ�飬���ı�I2��Һ25.00mL���ô�ʵ������Ʒ�п��������IJ�������������SO2���㣩Ϊ0.16g•L-1��
��������ʵ������У����в���HI��������������ⶨ���ƫ�ͣ��ƫ�ߡ���ƫ�͡����䡱����
���� ��1����װ�â��з����ķ�Ӧ��֪��װ�â��в���������ΪSO2���������������ᷴӦ���������ơ�����������ˮ��
��2��װ�â��л���������ľ��壬���������Һ̬��Ӧ��ȡ���˲�����
��3��װ�â����ڴ���β��������Ϊ��Ӧ�Ķ�������Ӧ��ֹ�������Ҳ��ܴ�����ȫ�ܱջ����У�
��4��NaHSO3��Һ��HSO3-�ĵ���̶ȴ���ˮ��̶ȣ���Һ�����ԣ��ʼ�����Һ�����Լ��ɣ�
��5��Na2S2O5�����ڿ������ױ�����ΪNa2SO4�������ᡢ�Ȼ�����Һ������Ʒ���Ƿ�����������ɣ�
��6���������ĵ���������SO2+I2+2H2O�TH2SO4+2HI��������������������������Ũ�ȣ�
�����в���HI�����������������ĵ����ƫС���ʲⶨ�����������ƫС��
��� �⣺��1����װ�â��з����ķ�Ӧ��֪��װ�â��в���������ΪSO2���������������ᷴӦ���������ơ�����������ˮ����Ӧ����ʽΪNa2SO3+H2SO4=Na2SO4+SO2��+H2O��
�ʴ�Ϊ��Na2SO3+H2SO4=Na2SO4+SO2��+H2O��
��2��װ�â��л���������ľ��壬���������Һ̬��Ӧ��ȡ���˽��з��룬�ʴ�Ϊ�����ˣ�
��3��a��װ��Ӧ���������백ˮ�п������ն�������Ϊ�ܱջ�����װ����ѹǿ�����ײ�����ȫ�¹ʣ��ʴ���
b����װ�����ն������������ϲ��Ϊ�ܱջ�����װ����ѹǿ�����ײ�����ȫ�¹ʣ��ʴ���
c����װ�ò������ն�������������ʵ��ʵ��Ŀ�ģ��ʴ���
d����װ���������������������Ӧ���������գ��ҷ�ֹ����������ȷ��
�ʴ�Ϊ��d��
��4��NaHSO3��Һ��HSO3-�ĵ���̶ȴ���ˮ��̶ȣ���Һ�����ԣ��ⶨ��Һ��pH������ȷ����Һ����ԣ�������Һ����ʹʪ����ɫʯ����ֽ��죬�������òⶨ��ҺpHֵ��ʪ�����ɫʯ����Һ���飬������Ba��OH��2��Һ��HCl��Һ��Ʒ����Һ������˵����Һ�����ԣ���ѡae��
�ʴ�Ϊ��ae��
��5��Na2S2O5��SԪ�صĻ��ϼ�Ϊ+4�ۣ���˻ᱻ����ΪΪ+6�ۣ��������ڿ������ױ�����ΪNa2SO4�������ᡢ�Ȼ�����Һ������Ʒ���Ƿ�����������ɣ�ʵ�鷽��Ϊ��ȡ����Na2S2O5�������Թ��У���������ˮ�ܽ⣬�μ����ᣬ���ٵμ��Ȼ�����Һ���а�ɫ�������ɣ�
�ʴ�Ϊ��ȡ����Na2S2O5�������Թ��У���������ˮ�ܽ⣬�μ����ᣬ���ٵμ��Ȼ�����Һ���а�ɫ�������ɣ�
��6������100mL���Ѿ��ж������������Ϊmg����
SO2+2H2O+I2�TH2SO4+2HI
64g 1mol
mg 0.025L��0.01mol/L
���ԣ�64g��mg=1mol��0.025L��0.01mol/L��
���m=0.016
�ʸô�ʵ������Ʒ�п��������IJ�������������SO2���㣩Ϊ$\frac{0.016g}{0.1L}$=0.16 g/L��
�ʴ�Ϊ��0.16��
�����в���HI�����������������ĵ����ƫС���ʲⶨ�����������ƫС����ⶨ���ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
���� ���⿼�����ʵ��Ʊ�ʵ�顢ʵ�鷽����ơ����ʺ����IJⶨ��������ԭ��Ӧ�ζ��ȣ��Ѷ��еȣ���ȷʵ��ԭ���ǽⱾ��ؼ����������ʵ����ʷ������ע��Ԫ�ػ�����֪ʶ�Ļ��ۺ�������ã�

| A�� | ���³�ѹ�£�18 gˮ�к��е�ˮ������ĿΪNA | |
| B�� | ���³�ѹ�£�1.06g Na2CO3���е�Na+������Ϊ0.02 NA | |
| C�� | ͨ��״���£�NA ��CO2����ռ�е����Ϊ22.4L | |
| D�� | ���ʵ���Ũ��Ϊ0.5mol/L��MgCl2��Һ�У�����Cl-����ΪNA |
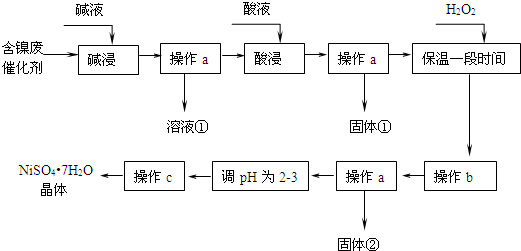
�������ܼʼ��������ȫ������pH��
| ������ | ��ʼ���� | ��ȫ���� |
| Al��OH��3 | 3.8 | 5.2 |
| Fe��OH��3 | 2.7 | 3.2 |
| Fe��OH��2 | 7.6 | 9.7 |
| Ni��OH��2 | 7.1 | 9.2 |
��1����Һ���к�����Ԫ�ص�������AlO2-��
��2�������ӷ���ʽ��ʾ����˫��ˮ��Ŀ��2Fe2++H2O2+2H+=2Fe3++2H2O��˫��ˮ���������������������A��
A������ B��ƯҺ C������ D������
��3������b������Һ��ΧΪ3.2-7.1����Ŀ���dz�ȥFe3+��
��4������a��c��Ҫ��ͬ�IJ��������Dz����������������У���ֹŨ���ᾧ������Ni2+ˮ��Ĵ�ʩ�ǵ�����ҺpHΪ2-3��
��5���������˫��ˮ���������ʱ��϶̡�����ʵ������Ӱ���Dz�Ʒ�л����̷������ʵ��֤����Ʒ���Ƿ����ʡ���������������Ӱ�죩��ȡ������Ʒ��������ˮ���μ����Ը��������Һ������Һ��ɫ��ȥ�����Ʒ�к���Fe2+��
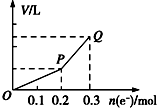 �ö��Ե缫�������ͭ��Һ����������ת�Ƶ��ӵ����ʵ������������������Ĺ�ϵ��ͼ��ʾ���������������ͬ״���²ⶨ������ʹ��Һ�ָ�����ʼ״̬��������Һ�м��루������
�ö��Ե缫�������ͭ��Һ����������ת�Ƶ��ӵ����ʵ������������������Ĺ�ϵ��ͼ��ʾ���������������ͬ״���²ⶨ������ʹ��Һ�ָ�����ʼ״̬��������Һ�м��루������| A�� | 0.15 mol CuO | B�� | 0.1 mol CuCO3 | ||
| C�� | 0.075mol Cu��OH��2 | D�� | 0.05 mol Cu2��OH��2CO3 |
| A�� | 1��2 | B�� | 2��3 | C�� | 3��2 | D�� | 1��1 |
| A�� | ���� | B�� | �ռ���Һ | C�� | AgNO3��Һ | D�� | KSCN��Һ |
