��Ŀ����

(2)���Ϊ�١��ڵ���������������__________________��______________��
(3)д�����������й�ҩƷ��ȡCl2�����ӷ���ʽ�� ____________________________��
 l2
l2(2)��Һ©����Բ����ƿ
(3)MnO2��4H����2Cl��
 Mn2����Cl2����2H2O
Mn2����Cl2����2H2O
ij�о���ѧϰС����������·����ⶨij���ҿ����м�ȩ�ĺ����������������������ԭ�����壩��
��.�ⶨԭ����KMnO4��H+����ҺΪǿ����������������ȩ�Ͳ��ᣬ�䷴Ӧ�����ӷ���ʽΪ��
4![]() +5HCHO+12H+====4Mn2++5CO2��+11H2O
+5HCHO+12H+====4Mn2++5CO2��+11H2O
2![]() +5H2C2O4+6H+====2Mn2++10CO2��+8H2O
+5H2C2O4+6H+====2Mn2++10CO2��+8H2O
��.�ⶨװ�ã�����װ������ͼ��ʾ��a��bΪֹˮ�У�

��.ʵ�鲽�裺
�ټ��װ�������ԣ����������ã���
����A����ȷ��ȡ25.00 mL 1.00��10-3 mol��L-1�ĸ��������Һ���������ڹ��ƿ�в�����3��6 mol��L-1 H2SO4��Һ���á�
�۽�2.00��10-3 mol��L-1�IJ������Һ����A�����б��á�
�ܴ�a���ر�b����ע������ȡ100 mL��װ�����ڿ������ر�a����b���ٻ����ƶ�ע������������ȫ������������������Һ�У�ʹ���ַ�Ӧ��������ظ�4�Σ���5�Σ���
�ݽ����ƿ�е���Һת����ƿ�У���ϴ���ƿ2��3�Σ�����ϴ��Һȫ��ת����ƿ����
���ñ�������Һ�ζ���ƿ�е���Һ����¼�ζ������ĵIJ�����Һ�����
�����ظ�ʵ��2�Ρ�
��.���ݴ������ֱ���װ���깤��ĵ�1�졢��7�졢��30�죨����ʼ�ձ���ͨ�绻��״���������ڿ�������ȡ����ͨ��ʵ���������������ݣ�ÿ��ʵ����ȡ��KMnO4��Һ��Ϊ25.00 mL����
װ���n�� | ������Һ/mL | ��ȩ��Ũ��/mg��m-3 | |||
1 | 2 | 3 | ƽ��ֵ | ||
1 | 15.86 | 15.72 | 15.67 | 15.75 |
|
7 | 26.24 | 26.36 | 26.31 | 26.30 |
|
30 | 30.90 | 30.67 | 30.81 | 30.79 |
|
������������⣺
��1��A����������_________________ ��
��2����������ѹ������ʱ�ٶȹ��죬���ܻ����ʲô���������
_____________________________________________________________________��
��3����������û����ϴ�������ȩ�ĺ�����____________���ƫ�ߡ���ƫ�͡�����Ӱ�족����
��4����ʵ���Ƿ���Ҫָʾ���������Ҫ����д��ָʾ�������ƣ��������Ҫ����˵���յ�ʱ��ʵ������___________________________________________________________________��
��5������KMnO4��Һ�����ʵ���Ũ��Ϊc1 mol��L-1����ȡKMnO4��Һ�����ΪV1 mL��������Һ�����ʵ���Ũ��Ϊc2 mol��L-1�����IJ�����Һ��ƽ�����ΪV2 mL��
�������ڿ����м�ȩŨ�ȣ�mg��m-3���Ĵ���ʽΪ____________����30�����ڿ����м�ȩŨ��Ϊ____________�����жϴ�ʱ����____________����ܡ�����ס��
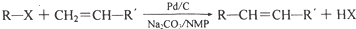

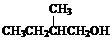
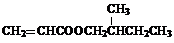 +H2O
+H2O
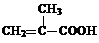



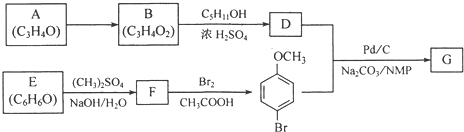
 �Ӳ�����B�������������Ǵ�����������������D�ǵؿ��к�����ߵ�Ԫ�أ�D��F��A��E�ֱ�ͬ���壻E�����ж���������Ԫ����ԭ�Ӱ뾶����Ԫ�ء�����������Ϣ�ش��������⣺
�Ӳ�����B�������������Ǵ�����������������D�ǵؿ��к�����ߵ�Ԫ�أ�D��F��A��E�ֱ�ͬ���壻E�����ж���������Ԫ����ԭ�Ӱ뾶����Ԫ�ء�����������Ϣ�ش��������⣺ _________________________��
_________________________�� C2D4(g)����H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ��
C2D4(g)����H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ�� 
 ______�㡣
______�㡣