��Ŀ����
��250C 101KPa�����µ��Ȼ��⣨HCl������49.6L��ͨ��492mLˮ�У�����֪ˮ���ܶ�Ϊ��1.00g/mL�����õ��ܶ�Ϊ1.13g/mL�����ᣨ���Ȼ����ˮ��Һ����ע��250C 101KPa����������Ħ�����Ϊ24.8��/mol��(����������һλС��)
��1����������������������5�֣�
��2�������������ʵ���Ũ�ȣ���3�֣�
��3��ȡ��20.0 mL����Һ�����Ũ��Ϊ1.00 mol/L��ϡ��Һ����ϡ�ͺ���Һ������Ƕ��ٺ�������3�֣�
��1��n(HCl)=2mol��1�֣�m(HCl)=73g��1�֣�m[HCl(aq)]=565g��1�֣�w=12.9%��2�֣�
��2��v[HCl(aq)]=500mL=0.5L��3�֣� c(HCl)= 4.0mol/L��3�֣�

��ϰ��ϵ�д�
�����Ŀ
 O2(g)�TCO2(g) ��H����283.0kJ/mol
O2(g)�TCO2(g) ��H����283.0kJ/mol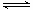 CO2(g)��H2(g) ��H��0�ں����ܱ������У���ʼʱn(H2O)=0.20mol��n(CO)��0.10 mol,��8000Cʱ�ﵽƽ��״̬��K��1.0����ƽ��ʱ��������CO��ת������_____________(����������һλС��)��
CO2(g)��H2(g) ��H��0�ں����ܱ������У���ʼʱn(H2O)=0.20mol��n(CO)��0.10 mol,��8000Cʱ�ﵽƽ��״̬��K��1.0����ƽ��ʱ��������CO��ת������_____________(����������һλС��)�� 2NH3(g)����H��0���ڵ��������½��У��ı�������Ӧ��������I��II��III����ϵ�и�����Ũ����ʱ��仯����������ͼ��ʾ��
2NH3(g)����H��0���ڵ��������½��У��ı�������Ӧ��������I��II��III����ϵ�и�����Ũ����ʱ��仯����������ͼ��ʾ��