��Ŀ����
�����£���amol N2��b mol H2�Ļ������ͨ��һ�̶��ݻ����ܱ������У��������·�Ӧ��N2(g)+3H2(g)  2NH3(g)��
2NH3(g)��
��1������Ӧ���е�ijʱ��tʱ�̣�n(N2)="13mol," n(NH3)=6mol,��a=?
��2����Ӧ�ﵽƽ��ʱ�������������Ϊ716.8L(���),����NH3�ĺ���Ϊ25%���������������ƽ��ʱn(NH3)=?
��3��ԭ��������У�a:b=?
��4��ƽ���������У�n(N2) :n(H2): n(NH3)=?
 2NH3(g)��
2NH3(g)����1������Ӧ���е�ijʱ��tʱ�̣�n(N2)="13mol," n(NH3)=6mol,��a=?
��2����Ӧ�ﵽƽ��ʱ�������������Ϊ716.8L(���),����NH3�ĺ���Ϊ25%���������������ƽ��ʱn(NH3)=?
��3��ԭ��������У�a:b=?
��4��ƽ���������У�n(N2) :n(H2): n(NH3)=?
��1��16mol ��2��8mol ��3��2�U3 ��4��3:3:2
������淴Ӧ���йؼ��㡣
��1��������6mol���������ĵ�����3mol�����a��3mol+13mol��16mol��
��2����״����716.8L������ ����������32mol��0.25��8mol��
����������32mol��0.25��8mol��
��3��ƽ��ʱ���ɰ�����8mol�������ĵ�����4mol������12mol������ƽ��ʱ���������������32mol��12mol��8mol��12mol������b��12mol��12mol��24mol������a:b��16�U24��2�U3��
��4�����ݣ�3����֪ƽ���������У�n(N2) :n(H2): n(NH3)��12�U12�U8��3:3:2��
��1��������6mol���������ĵ�����3mol�����a��3mol+13mol��16mol��
��2����״����716.8L������
 ����������32mol��0.25��8mol��
����������32mol��0.25��8mol����3��ƽ��ʱ���ɰ�����8mol�������ĵ�����4mol������12mol������ƽ��ʱ���������������32mol��12mol��8mol��12mol������b��12mol��12mol��24mol������a:b��16�U24��2�U3��
��4�����ݣ�3����֪ƽ���������У�n(N2) :n(H2): n(NH3)��12�U12�U8��3:3:2��

��ϰ��ϵ�д�
 ��һ������ĩ�ٷֳ�̾�ϵ�д�
��һ������ĩ�ٷֳ�̾�ϵ�д�
�����Ŀ
 2C��������2min��Ӧ�ﵽƽ��ʱ����û�����干3.4 mol������0.4 mol C�������м���������ȷ����
2C��������2min��Ӧ�ﵽƽ��ʱ����û�����干3.4 mol������0.4 mol C�������м���������ȷ����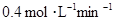
 2NH3(g)����H<0��673K��30MPa��n(NH3)��n(H2)��ʱ��仯�Ĺ�ϵ����ͼ��ʾ������������ȷ����
2NH3(g)����H<0��673K��30MPa��n(NH3)��n(H2)��ʱ��仯�Ĺ�ϵ����ͼ��ʾ������������ȷ����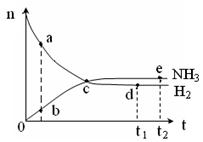
 cC(g)+dD(g)��ƽ������¶Ȳ��䣬���������ѹ����ԭ����һ�롣���ﵽ�µ�ƽ��ʱ��D��Ũ��Ϊԭƽ��ʱ��1.6������
cC(g)+dD(g)��ƽ������¶Ȳ��䣬���������ѹ����ԭ����һ�롣���ﵽ�µ�ƽ��ʱ��D��Ũ��Ϊԭƽ��ʱ��1.6������ pC(g)����ƽ����¶�һ��ʱ�������������Сԭ����1/2�����ﵽ�µ�ƽ��ʱ��C��Ũ��Ϊԭ����1.9������ѹ�������б����¶Ȳ��䣬��Ӧ����ʽ������ϵ����ϵ��
pC(g)����ƽ����¶�һ��ʱ�������������Сԭ����1/2�����ﵽ�µ�ƽ��ʱ��C��Ũ��Ϊԭ����1.9������ѹ�������б����¶Ȳ��䣬��Ӧ����ʽ������ϵ����ϵ�� 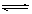 B(g) + D(g)�����ֲ�ͬ�����½��У���Ӧ����Ϊ��ͬ�����ܱ�������B��D��ʼŨ��Ϊ0����Ӧ��A��Ũ�ȣ�
B(g) + D(g)�����ֲ�ͬ�����½��У���Ӧ����Ϊ��ͬ�����ܱ�������B��D��ʼŨ��Ϊ0����Ӧ��A��Ũ�ȣ�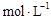 ���淴Ӧʱ�䣨min���ı仯������±���
���淴Ӧʱ�䣨min���ı仯������±��� 2C(g)�ﵽƽ��״̬�ı�־�ǣ� ��
2C(g)�ﵽƽ��״̬�ı�־�ǣ� ��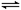 xC(g)��2D(g)��2 minĩ�÷�Ӧ�ﵽƽ�⣬����0.8 mol D�������C��Ũ��Ϊ0.2 mol��L��1�������ж���ȷ����
xC(g)��2D(g)��2 minĩ�÷�Ӧ�ﵽƽ�⣬����0.8 mol D�������C��Ũ��Ϊ0.2 mol��L��1�������ж���ȷ����