��Ŀ����
ֱ������̼��̼���ķ�Ӧ��ʵ�ָ�Ч����ɫ�л��ϳɵ���Ҫ;������������ż����Ӧ�ǽ��걸�ܹ�ע��һ��ֱ������̼��̼�����·�Ӧ�����磺
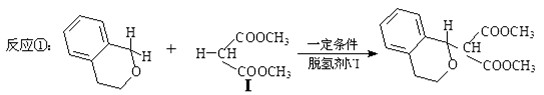
�������������ºϳ�·��ã�
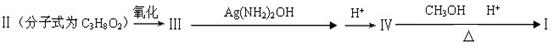
��1���������ķ���ʽΪ____________������ȫˮ��Ļ�ѧ����ʽΪ_____________��ע����������
��2���������������Ũ�����ᷴӦ�Ļ�ѧ����ʽΪ_____________��ע����������
��3���������û�����ԣ���ṹ��ʽΪ____________�����һ��ͬ���칹������뱥��NaHCO3��Һ��Ӧ�ų�CO2����������Ľṹ��ʽΪ___________________��
��4����Ӧ����1������������ṹ��ʽ���£����ӻ��2����ԭ�Ӻ�ת���1�������廯������ӣ��÷����廯������ӵĽṹ��ʽΪ_________________��
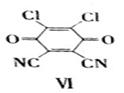
��5��1���� ��1����
��1���� ��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ���������ӵĽṹ��ʽΪ____________��1mol�ò���������______molH2�����ӳɷ�Ӧ��
��һ�������¿ɷ������Ʒ�Ӧ�ٵķ�Ӧ���������ӵĽṹ��ʽΪ____________��1mol�ò���������______molH2�����ӳɷ�Ӧ��
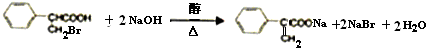
��24�֣���1��C5H8O4��H3COOCCH2COOCH3+2NaOH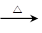 2CH3OH+NaOOCCH2COONa��
2CH3OH+NaOOCCH2COONa��
��2��HOCH2CH2CH2OH+2HBr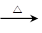 CH2BrCH2CH2Br+2H2O��
CH2BrCH2CH2Br+2H2O��
��3��OHCCH2CHO��CH2��CHCOOH��
��4��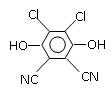 ����5��
����5��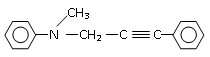 ��8��
��8��
��������
�����������1�����ݻ�����I�Ľṹ��ʽ��֪���û�����Ļ�ѧʽӦ����C5H8O4�������к���2��������Ҫ��ȫˮ�⣬��Ӧ���ڼ��������½��У�������ȫˮ��Ļ�ѧ����ʽΪ
H3COOCCH2COOCH3+2NaOH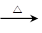 2CH3OH+NaOOCCH2COONa��
2CH3OH+NaOOCCH2COONa��
��2����������������ɻ�����I�����Ը��ݻ�����I�Ľṹ��ʽ���жϣ��������Ӧ����1��3�����������ܺ������ᷢ��ȡ����Ӧ������ʽ��HOCH2CH2CH2OH+2HBr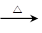 CH2BrCH2CH2Br+2H2O��
CH2BrCH2CH2Br+2H2O��
��3����������������ɻ�������һ������û�����ԣ��ܷ���������Ӧ�����Ի������Ľṹ��ʽΪOHCCH2CHO�����һ��ͬ���칹������뱥��NaHCO3��Һ��Ӧ�ų�CO2����˵�������к����Ȼ������Ի�������Ľṹ��ʽΪCH2��CHCOOH��
��4��1������������ӻ��2����ԭ�Ӻ�ת���1�������廯������ӣ�����б������ɣ����Ը���ԭ���غ��֪���÷����廯������ӵĽṹ��ʽΪ ��
��
��5��������֪����Ϣ��֪���ڷ�Ӧ������ȥ��1��������������������Ľṹ��ʽ��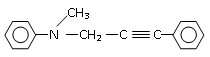 �����ݽṹ��ʽ��֪������2��������1��̼̼����������1mol�ò���������8molH2�����ӳɷ�Ӧ��
�����ݽṹ��ʽ��֪������2��������1��̼̼����������1mol�ò���������8molH2�����ӳɷ�Ӧ��
���㣺�����л������ʽ���ṹ��ʽ���жϡ�ͬ���칹����ж��Լ���ѧ����ʽ����д��
����������л��ϳ���Ŀ�Ĺؼ����ڣ���ѡ��������ĺϳ�·�ߣ����������պø����л������ɡ��ṹ�����ʡ��������ϵ�Լ���Ҫ�����ŵ���������ȥ�Ȼ���֪ʶ��

 ���ٴ�����ɽ����ϵ�д�
���ٴ�����ɽ����ϵ�д�| �ṹ��ʽ |  |  |  |
| ����ʽ |  |  |  |
��2��
 �ķ���ʽΪ______��
�ķ���ʽΪ______����3����Ȳ3���ӾۺϵIJ���Ϊ����д����Ȳ�����Ӿۺ��γɵķ��������ӵļ���ʽ______
��4����
 �Լ���Ҫ�����Լ��ɺϳ�
�Լ���Ҫ�����Լ��ɺϳ� ��д����һ���ϳɷ�Ӧ�Ļ�ѧ����ʽ______��
��д����һ���ϳɷ�Ӧ�Ļ�ѧ����ʽ______����5��
 ��һ��ͬ���칹��A������һ�ȴ��������֣���Br2��1��1�ӳɲ���ֻ��һ�֣�������˳���������칹������A�Ľṹ��ʽΪ______��
��һ��ͬ���칹��A������һ�ȴ��������֣���Br2��1��1�ӳɲ���ֻ��һ�֣�������˳���������칹������A�Ľṹ��ʽΪ______�����һ����ȩ��
 ����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����
����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ������1���һ����ȩ�����еĺ�������������������______��______���˴Ź����ױ����÷�������______�ֲ�ͬ���͵���ԭ�ӣ�
�һ����ȩ��ͬ���칹��A��һ���л��ᣬA�ɷ������±仯��

��ʾ��a��RCH2OH
 RCHO
RCHOb���뱽��ֱ��������̼ԭ��������ʱ����̼ԭ�Ӳſɱ�����KMnO4��Һ����Ϊ�Ȼ���
��2��A����C�ķ�Ӧ������______��
��3��д���ڼ���������C��NaOHˮ��Һ������Ӧ�Ļ�ѧ����ʽ______��
��4���һ����ȩ����һ��ͬ���칹��D��
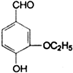
 ����һ��ҽҩ�м��壮
����һ��ҽҩ�м��壮����ƺ�������������ȩ��
 ���ϳ�D���÷�Ӧ����ͼ��ʾ���£��뽫����һ���ķ�Ӧ����д�ڼ����·����Լ���ԭ��д�ڼ����Ϸ����м����������ͼ�еķ����ڣ���д������ȩ��������Һ��Ӧ�Ļ�ѧ����ʽ��
���ϳ�D���÷�Ӧ����ͼ��ʾ���£��뽫����һ���ķ�Ӧ����д�ڼ����·����Լ���ԭ��д�ڼ����Ϸ����м����������ͼ�еķ����ڣ���д������ȩ��������Һ��Ӧ�Ļ�ѧ����ʽ�� ______��______��
______��______��





 �ķ���ʽΪ
�ķ���ʽΪ
 �Լ���Ҫ�����Լ��ɺϳ�
�Լ���Ҫ�����Լ��ɺϳ� ��д����һ���ϳɷ�Ӧ�Ļ�ѧ����ʽ
��д����һ���ϳɷ�Ӧ�Ļ�ѧ����ʽ
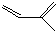 ��һ��ͬ���칹��A������һ�ȴ��������֣���Br2��1��1�ӳɲ���ֻ��һ�֣�������˳���������칹������A�Ľṹ��ʽΪ
��һ��ͬ���칹��A������һ�ȴ��������֣���Br2��1��1�ӳɲ���ֻ��һ�֣�������˳���������칹������A�Ľṹ��ʽΪ
 ����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����
����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����

 ����һ��ҽҩ�м��壮
����һ��ҽҩ�м��壮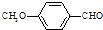 ���ϳ�D���÷�Ӧ����ͼ��ʾ���£��뽫����һ���ķ�Ӧ����д�ڼ����·����Լ���ԭ��д�ڼ����Ϸ����м����������ͼ�еķ����ڣ���д������ȩ��������Һ��Ӧ�Ļ�ѧ����ʽ��
���ϳ�D���÷�Ӧ����ͼ��ʾ���£��뽫����һ���ķ�Ӧ����д�ڼ����·����Լ���ԭ��д�ڼ����Ϸ����м����������ͼ�еķ����ڣ���д������ȩ��������Һ��Ӧ�Ļ�ѧ����ʽ��


 ����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����
����ʳƷ���Ӽ�������ԭ�ϣ�����ζ�����ȩ����Ũ����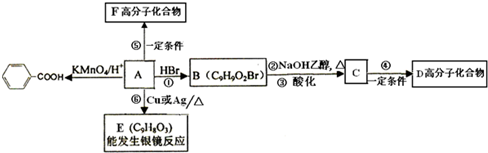

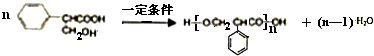
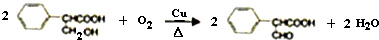


















 ͨ������£�����ǻ�����ͬһ��̼ԭ���ϵķ��ӽṹ�Dz��ȶ��ģ������Զ�ʧˮ������̼��˫���Ľṹ��
ͨ������£�����ǻ�����ͬһ��̼ԭ���ϵķ��ӽṹ�Dz��ȶ��ģ������Զ�ʧˮ������̼��˫���Ľṹ��