��Ŀ����
15�� ijѧ����0.1000mol/L KOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ������ɷֽ�Ϊ���¼�����
ijѧ����0.1000mol/L KOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ������ɷֽ�Ϊ���¼�������A����ʢ�б���Һ�ļ�ʽ�ζ��̶ܹ��ã�����Һ��ʹ�ζ��ܼ��������Һ��
��B���ñ���Һ��ϴ�ζ���2-3�Σ�
��C����ȡ20.00mL�����������Һע��ྻ����ƿ��������2-3�η�̪��
��D������Һ����0��0�̶����£����¶�����Ϊ4.60mL��
��E��ȡ��KOH��Һע���ʽ�ζ�����0�̶�����2-3cm��
��F������ƿ���ڵζ��ܵ����棬����һ�Ű�ֽ���ñ�KOH��Һ�ζ� ���յ㣬���µζ���Һ��Ŀ̶ȣ����ζ�����ʱ���ζ���Һ����ͼ��ʾ�����յ����Ϊ22.60mL��
���������գ�
��1����ȷ������˳���ǣ��������ĸ��д����B����E����A����D����C����F����
��2��ʵ���������ֿ��Ƽ�ʽ�ζ�����Ƥ�ܲ����鴦������������λ�����۾�ע����ƿ����Һ����ɫ�仯��ֱ���ζ��յ㣮�жϵ����յ����������ƿ����Һ����ɫ����ɫ��Ϊ�ۺ�ɫ�ұ��ְ���Ӳ���ɫ��
��3��������A������֮ǰ�������ô���Һ��ϴ��ƿ����Բⶨ�����Ӱ���ǣ�ѡ����ţ���ƫ��ƫС������Ӱ�죬��ͬ���٣�����ȡһ������KOH���壨������NaOH�����Ʊ���Һ�������ζ��������ᣬ��Բⶨ�����Ӱ���Ǣڣ����������У���ȱ�٣�B����������Եζ������Ӱ���Ǣ٣��ζ��������縩�ӹ۲�ζ�����Һ��̶ȣ���Եζ������Ӱ���Ǣڣ� ���ݶ�����ʽ������ô���Һ��pH1.0458��1.046�����������ϸ������������֪��lg2=0.3010��lg3=0.4771��lg5=0.6990��
���� ��1���к͵ζ��м�©��ϴ�ӡ���ϴ��װҺ���ζ��Ȳ�����
��2��ʵ�������ֿ��Ʋ����飬Ӧ�������ٲ��۲���ƿ����ɫ�仯������Һ��ɫ�仯�Ұ�����ڲ���ɫ����˵���ﵽ�ζ��յ㣻
��3������c�����⣩=$\frac{c��������V������}{V�����⣩}$���������ݹ�ϵʽHCl��KOH������������Ũ�ȣ�
��� �⣺�ζ��� 0�̶����Ϸ�������ͼ��֪�յ����Ϊ22.60mL���ʴ�Ϊ��22.60��
��1���к͵ζ����ռ�©��ϴ�ӡ���ϴ��װҺ��ȡ����Һ����ָʾ�����ζ���˳�����������ȷ��˳��ΪBEADCF���ʴ�Ϊ����B����E����A����D����C����F����
��2���ζ�ʱ����Ӧ���Ƽ�ʽ�ζ��ܵIJ����飬�Ա����Һ������٣�����ҡ����ƿ������Ӧ��ע����ƿ����Һ����ɫ�仯���ζ�ʱ����ƿ����Һ����ɫ����ɫ��dz���ұ��ְ�����ڲ���ɫ����˵���ﵽ�ζ��յ㣬
�ʴ�Ϊ����ʽ�ζ�����Ƥ�ܲ����鴦����ƿ����Һ����ɫ����ɫ��Ϊ�ۺ�ɫ�ұ��ְ���Ӳ���ɫ��
��3��������A������֮ǰ�������ô���Һ��ϴ��ƿ������Һ�����ʵ���ƫ�������ĵı�Һ���ƫ����c�����⣩=$\frac{c��������V������}{V�����⣩}$����֪c�����⣩ƫ��
����NaOH��Ħ������С��KOH������KOH�к���������NaOH������c������ƫ��V������ƫС���ʴ���Һ��Ũ��ƫС������ϴ�ζ��ܣ����±���Һ��ϡ�ͣ�V������ƫ����ô����Һ��Ũ��ƫ�ζ��������縩�ӹ۲�ζ�����Һ��̶ȣ���ôV�����⣩ƫ����ô����Һ��Ũ��ƫС��
���ı�KOH��Һ�����Ϊ22.60mL-4.60mL=18.00mL��
HCl��KOH
1 1
c��HCl����20.00mL 0.1000mol•L-1��18.00mL��
c��HCl��=$\frac{0.1000mol/L��18.00mL}{20.00mL}$=0.09000mol•L-1��pH=-log0.09=-��-2+2log3��=2-2��0.4771=1.0458��
�ʴ�Ϊ���٣��ڣ��٣��ڣ� 1.0458��1.046��
���� ���⿼���к͵ζ�����ȷ�ζ�ʵ���е����������ݴ������к͵ζ����������ǽ����Ĺؼ���ע������к͵�ʵ�ʣ���Ŀ�Ѷ��еȣ�

| A�� | ��Զ���֬���ȶ��ֲ�������� | |
| B�� | ��֬������ֱ�������������� | |
| C�� | ��֬��Ϊ��̬ | |
| D�� | ��Щ֬�������������ģ���������ȴ���ܺϳ� |
 ������㷺����ҽҩ��ʳƷ��Ⱦ�ϡ�����������������ʵ�����Ʊ��������һ�ַ�������Ӧԭ�����£�
������㷺����ҽҩ��ʳƷ��Ⱦ�ϡ�����������������ʵ�����Ʊ��������һ�ַ�������Ӧԭ�����£� +2KMnO4$\stackrel{��}{��}$
+2KMnO4$\stackrel{��}{��}$ +2MnO2��+KOH+H2O
+2MnO2��+KOH+H2O +HCl��
+HCl��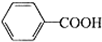 ��+KCl
��+KCl������ϣ�
| ���� | ��Է������� | ��״ | �۵�/�� | �ܽ�� | |
| ˮ | �Ҵ� | ||||
| �ױ� | 92 | ��ɫҺ����ȼ�ӷ� | -96 | ���� | ���� |
| ������ | 122 | ��ɫƬ״����״���� | 122.4 ��100�������� | 25��0.35g 80��2.7g | ���� |
��ͼ��װ��Ӧװ�ã�����ˮ���гּ�����װ��δ������������������ƿ�����μ���120mLˮ��9��.4g������غ�3.0mL�ױ���
�ڽ�������ƿ�еĻ��Һ����裬���������ڣ�ֱ���ױ���ȫ��Ӧ��
�۳��ȹ��˷�Ӧ��������Һ����ɫ���������������������������Һ����ɫ��ȥ���ٹ��ˣ�����ˮϴ��������ϴ��Һ�ϲ�����Һ�У�
���ñ�ˮ��ȴ��Һ��Ȼ����Ũ�����ữ�����ˣ���������ˮϴ���������õ�������ֲ�Ʒ�����ؽᾧ�õ����Ƶı����ᣮ
�ش��������⣺
��1����ʵ��Ӧѡ���������ƿ���ΪB������ĸ��ţ���
A��100mL B��250mL C��500mL D��1000mL
��2���жϼױ�����ȫ��Ӧ��������������ƿ�л��Һ���ٷֲ㡢����Һ���ٳ������飮
��3��ʵ�鲽����У���������ˮ��������ˮϴ��������Ŀ���Ǽ��ٱ��������ܽ������ʧ��������ֲ�Ʒ���˿����ؽᾧ�������⣬������B����
A������ B������ C������ D����Һ
��4�����Ƶı����ᴿ�Ȳⶨ����ȡ1.220g��Ʒ����ϡ�Ҵ��ܽⲢ���100mL��Һ���ֱ�ȡ25��.00mL��Һ����0.1000mo1•L-1NaOH����Һ�ζ������εζ�����NaOH��Һ������ֱ�ΪV1=24.70mL��V2=24.80mL��V3=25.80mL��
��������Һʱ��ϡ�Ҵ�����������ˮ���ܼ���ԭ���dz����±���������ˮ�������Ҵ���
�����÷�̪��ָʾ����ȷ���ζ��յ�������ǵμ����һ���������Ʊ���Һ����Һ����ɫ��Ϊdz��ɫ����30�벻��ɫ��
�۲�Ʒ�Ĵ���Ϊ99.00%��
| X | Y | Z | M | R | Q | |
| ԭ�Ӱ뾶/nm | 0.186 | 0.074 | 0.099 | 0.143 | ||
| ��Ҫ���ϼ� | -4��+4 | -2 | -1��+7 | +3 | ||
���� | �����Ӻ������� | �����뵼����� | ��ɫ��Ӧ�ʻ�ɫ |
 ��R����Ȼ������������Ϊ35��37�����ֺ��أ�����֮��Ĺ�ϵ��Ϊͬλ�أ�
��R����Ȼ������������Ϊ35��37�����ֺ��أ�����֮��Ĺ�ϵ��Ϊͬλ�أ���2��MԪ�صij��������ж��֣����ǻ���ͬ�������壬���ǵĻ�ѧʽΪO2��O3��
��3��Z�ĵ�����ˮ��Ӧ�Ļ�ѧ����ʽ��2Na+2H2O=2NaOH+H2����
��4��Y��R��ȣ��ǽ����Խ�ǿ��Ԫ����Cl��������ʵ��֤����һ���۵���BC��ѡ����ĸ��ţ���
A��������Y�ĵ��ʳʹ�̬��R�ĵ��ʳ���̬����B���ȶ���XR��YX4
C��Y��R�γɵĻ�������Y�����ۡ�����������D����ˮ�е��ܽ��ԣ�X4YM4��XRM4
��5�����������Ӧ��������ڲ�ͬ�ܼ��Ĺ����ԾǨ�йص���ABD��
A��Xԭ�������ֿɼ��������� ��B��ZԪ����ɫ��Ӧ�ʻ�ɫ
C��Y���ʿ����뵼����� �� ��D��Q�������������ڸ����¾��ҷ�Ӧ�����⡢���ȣ�
| A�� | 1mol������1mol��������ȫ��Ӧ��Ҫ����26kJ������ | |
| B�� | 1������Ӻ�1���������ȫ��Ӧ��Ҫ����52kJ������ | |
| C�� | 1mol H2��g����1mol I2��g����ȫ��Ӧ����2mol��HI��g��������52kJ������ | |
| D�� | 1mol H2��g����1mol I2��g����ȫ��Ӧ�ų�26kJ������ |

 ��֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵�����������ӣ�����A��B��A��D�����ڱ���λ�����ڣ�Aԭ�Ӻ���������δ�ɶԵ��ӣ�BԪ�صĵ�һ�����ܱ�ͬ������������Ԫ�ض���Cԭ����ͬ����ԭ���а뾶���ϡ��������⣩��E��Cλ�ڲ�ͬ���ڣ�Eԭ�Ӻ���������������C��ͬ�����������Ӿ������������������Ϣ���ش��������⣺������ʱA��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��
��֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵�����������ӣ�����A��B��A��D�����ڱ���λ�����ڣ�Aԭ�Ӻ���������δ�ɶԵ��ӣ�BԪ�صĵ�һ�����ܱ�ͬ������������Ԫ�ض���Cԭ����ͬ����ԭ���а뾶���ϡ��������⣩��E��Cλ�ڲ�ͬ���ڣ�Eԭ�Ӻ���������������C��ͬ�����������Ӿ������������������Ϣ���ش��������⣺������ʱA��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��