��Ŀ����
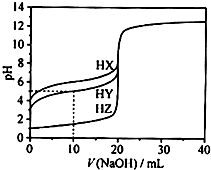
25��ʱ����Ũ��Ϊ0.1000 mol��L��1��NaOH��Һ�ζ�20.00 mLŨ�� ��Ϊ0.1000 mol��L��1��������HX��HY��HZ�ζ�������ͼ��ʾ������˵����ȷ����

A������ͬ�¶��£�ͬŨ�ȵ���������Һ�ĵ�������˳��HZ��HY��HX
B�����ݵζ����ߣ��ɵ�Ka(HY)��10��5
C��������HX��HY��Һ�������Ϻ���NaOH��Һ�ζ���HXǡ����ȫ��Ӧʱ��
c(X��)��c(Y��)��c(OH��)��c(H��)
D��HY��HZ��ϣ��ﵽƽ��ʱ��c(H��)�� ��c(Z��)��c(OH��)
��c(Z��)��c(OH��)
B
��������
���������Ũ�Ⱦ�Ϊ0.1000 mol/L��������HX��HY��HZ�����ݵζ�����0���������pH�ɵõ�HZ��
ǿ�ᣬHY��HX�����ᣬ�����ԣ�HY��HX��Aѡ�ͬ��ͬŨ��ʱ��������ĵ����ԣ�HZ��HY��
HX��Bѡ���NaOH��Һ�μӵ�10 mLʱ����Һ��c(HY)��c(Y��)����Ka(HY)��c(H+)=10��pH=10��5��
Cѡ���NaOH��Һ�ζ���HXǡ����ȫ��Ӧʱ��HY�类��ȫ�кͣ�������Һ��NaY��NaX���
��Һ���������ԣ�HY��HX����X����ˮ��̶ȴ���Y������Һ��c(Y��)��c(X��)��Dѡ�HY��HZ��
�ϣ���Һ�ĵ���غ�ʽΪ��c(H+)= c(Y��)+ c(Z��) +c(OH��)���ָ���HY�ĵ���ƽ�ⳣ����
Ka(HY)= ����c(Y��)=
����c(Y��)=
���Դ�ƽ���c(H+)= + c(Z��) +c(OH��)
+ c(Z��) +c(OH��)
���㣺������Һ��������⣬�漰��pHֵ����Һϡ�͵�������⡣

 �ƸԹھ��ο���ϵ�д�
�ƸԹھ��ο���ϵ�д����ݱ�� |
NaOH��������/mL |
��Һ��pH | ||
| C��HX�� | c��HY�� | |||
| �� | 0 | 3 | 1 | |
| �� | a | 7 | ||
| �� | 20.00 | x | y | |
| A������ͬ�¶��£�ͬŨ�ȵ���������Һ�ĵ�������˳��HX��HY |
| B���ɱ������ݿɹ����Ka��HX����10-5 |
| C��HY��HX��ϣ��ﵽƽ��ʱ���������غ� |
| D�������ڷ�Ӧ���HY��Һ�У�c��Na+����c��Y-����c��OH-����c��H+�� |
 ��2013?�㽭��25��ʱ����Ũ��Ϊ0.1000mol?L-1��NaOH��Һ�ζ�20.00mLŨ�Ⱦ�Ϊ0.1000mol?L-1��������HX��HY��HZ�ζ�������ͼ��ʾ������˵����ȷ���ǣ�������
��2013?�㽭��25��ʱ����Ũ��Ϊ0.1000mol?L-1��NaOH��Һ�ζ�20.00mLŨ�Ⱦ�Ϊ0.1000mol?L-1��������HX��HY��HZ�ζ�������ͼ��ʾ������˵����ȷ���ǣ�������
 ��c(Z��)��c(OH��)
��c(Z��)��c(OH��)