��Ŀ����
����˵����ȷ����
A��NaHCO3��Na2CO3�����Һ�У�һ����
c(Na��)��c(H��)��c(OH��)��c(HCO3-) ��c(CO32-)
B��Ũ�Ⱦ�Ϊ0.1 mol��L��1��������Һ��pH�ɴ�С����˳��Ϊ
NaOH>Na2CO3>NaHSO4>(NH4)2SO4
C�������������μ�ˮ����Һ�ĵ����ԡ�����ĵ���̶ȡ�pH����������С
D��pH=3�������������Һ��c(SO42��)��c(CH3COO��)֮��Ϊ1��2
D
��������
���������A������ȷ������غ�ӦΪ��c(Na��)��c(H��)��c(OH��)��c(HCO3-) ��2c(CO32-)��B������ȷ��NaOH��ǿ����ʣ���ȫ���룬c��OH�� ��=0.1mol��L��1������pH=13��Na2CO3 ��ǿ�������Σ�ˮ��Һ���ʼ��ԣ�pHС��13��(NH4)2SO4��ǿ�������Σ�ˮ�����Һ�����ԣ�1��pHֵ��7��NaHSO4��ǿ����ʽ�Σ���ˮ����ȫ����������ӡ���������ӡ������ӣ�����C��H����=0.1mol��L��1������pH=1������pH�ɴ�С����˳��ΪNaOH>Na2CO3>(NH4)2SO4>NaHSO4��C������ȷ���ڱ���������μ�ˮ���������ܽⲢ���룬����̶�����������Ũ�����������ڣ���������ϡ�ͣ�����Ũ�ȼ�С������Һ�ĵ����Խ���С������̶�����Һ��ϡ�ͣ�Խ��Խ����Һ��pH��������С��D����ȷ�� pH=3��H2SO4��c(H�� )=0.001mol/L,c(SO42�D )=0.0005mol/L,C(CH3COO�D)=0.001mol/L��c(SO42��)��c(CH3COO��)֮��Ϊ1��2��ѡD��
���㣺�����Һ

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�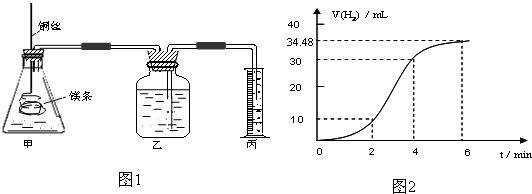
��1��ʵ��1 ̽��Mg�����ᷴӦ���ʵı仯���ɣ�ȡһ��þ������ɰֽ��ȥ���������Ĥ��ͭ˿����þ������װ�ü��У�ʹþ��������ƿ�ڵ����Ϊ2Lϡ���ᣨ�������У�þ�������ᷴӦ����H2������뷴Ӧʱ��Ĺ�ϵ������ͼ2��ʾ��
�ٴ�ͼ2�п���0-6min��ƽ����Ӧ��������ʱ�����
A��0-2min B��2-4min C��4-6min
�������4-6min ʱ���ڣ���HCl��ʾ��ƽ����Ӧ����Ϊ
��ͼ1װ�ü�����þ��������ͭ˿��һ�����ϡ�����жԷ�Ӧ����Ӱ������˵����ȷ����
A���ӿ췴Ӧ���ʵ������������������� B��������Ӧ������������������
C����Ӱ�췴Ӧ���� D���ӿ췴Ӧ���ʵ�����������������С
��2��ʵ��2 ̽����Ũ�ȶ�MnO2��H2O2��Ӧ���ʵ�Ӱ��
��֪MnO2+H2O2+2H+�TMn2++O2��+2H2O����ȡ����MnO2���±��й����ʣ�����ͬ�¶��½���4��ʵ�飬�ֱ��¼�ռ�20.0mL��������ʱ�䣮
| ʵ���� | �� | �� | �� | �� |
| 10%H2O2�����/mL | 5.0 | 5.0 | V1 | V2 |
| 20%��������/mL | 0 | 0.5 | 1.0 | V3 |
| ˮ�����/mL | 15 | 14.5 | V4 | 13.5 |
| ����ʱ��t/s | t1 | t2 | t3 | t4 |
����ͬѧ���ʵ��I������Ϊʵ����ĶԱ�ʵ�飬��������
����ʵ����t2��t3��t4����ɵó���ʵ�������


 �ǽ���Ԫ�ص��ж����������NO��NO2��N2O4�ȣ�
�ǽ���Ԫ�ص��ж����������NO��NO2��N2O4�ȣ�