��Ŀ����
����ɸ������������ǿ�����ȶ��Ըߵ��������ܣ�ʹ�÷���ɸ��ù㷺Ӧ�ã�ij���ͺŷ���ɸ�Ĺ�ҵ�������̿ɱ�ʾ���£�
�ڵμӰ�ˮ����pH=9�Ĺ����У���pH���Ʋ���������Al(OH)3���ɣ�����������������Ԫ�ء���Ԫ�ؾ�û����ʧ����ԭ��������Ϊ10%��
��1������ɸ��ֱ��Ϊ4A(1A=10��10m)��Ϊ4A�ͷ���ɸ����Na+��Ca2+ȡ��ʱ���Ƶ�5A�ͷ���ɸ����Na+��K+ȡ��ʱ���Ƶ�6A�ͷ���ɸ��Ҫ��Ч����������(����ֱ��Ϊ4��65A)���춡��(����ֱ��Ϊ5��6A)Ӧ��ѡ��_______�ͷ���ɸ��
��2��Al2(SO4)3��Һ��Na2SiO3��Һ��Ӧ���ɽ�������ӷ���ʽΪ________________________
��3��������������������Һ�ﺬ�е����ӳ�H+��OH���⣬��Ҫ���� �����ӡ��������н��������ӵIJ��������� ��
��4����NH3?H2O����pH���ȵ�90�沢���ȹ��˵�ԭ������� ��
��5�����������������÷���ɸ�Ļ�ѧʽΪ ��������������ʽ��ʾ��
��1��5A��2��2Al3++3SiO32-+6H2O=2Al(OH)3+3H2SiO3
��3��Na+,NH4+,SO42-;����˿������˿���ھƾ����ϼ�������ɫ��պȡ������Һ���ھƾ������������գ�������ʻ�ɫ�������Һ�к���Na+;
��4�������ܴ�ʹ�������ۣ����ȹ��˿ɷ�ֹ�������ʾ���������
��5��Na2O��Al2O3��10SiO2��6H2O
���������������1������������ķ���ֱ��Ϊ4��65A���춡��ķ���ֱ��Ϊ5��6A������Ӧ��ѡ��5A�ͷ���ɸ�ſ��Խ����߷��뿪����2��Al2(SO4)3��ǿ�������Σ� Na2SiO3��ǿ�������Σ���������Һ��Ӧ���ɽ�������ӷ���ʽΪ2Al3++3SiO32-+6H2O=2Al(OH)3+3H2SiO3����3�����������غ㶨�ɣ���֪�ڸ�����������������Һ�ﺬ�е����ӳ�H+��OH���⣬��Ҫ����Na+��NH4+��SO42-����������ɫ��Ӧ���������н���������Na+��������������ǽ���˿������˿���ھƾ����ϼ�������ɫ��պȡ������Һ���ھƾ������������գ����۲쵽����ʻ�ɫ�������Һ�к���Na+����4����NH3?H2O����pH���ȵ�90�沢���ȹ�������Ϊ�����ܴ�ʹ�������ۣ����ȹ��˿ɷ�ֹ�������ʾ������������½��岻������5��������������������Ԫ�ء���Ԫ�ؾ�û����ʧ����ԭ��������Ϊ10%��n(Al)=(34��2g��342g/mol)��2=0��2mol��n(Si)=122g��122g/mol=1mol��n(Na)=2n(Si)=2mol�������ڽ����е�Al:0��2mol;Si:1mol;Na: 2mol��10%=0��2mol;�����ڻ������е��������ϼ۵Ĵ�����Ϊ0������2n(O)=0��2��3+1��4��0��2��1�����n(O)=2��4������n(Na): n(Al): n(Si): n(O)=0��2:0��2:1:2��4=1:1:5:12�����ڷ���ɸ�Ļ�ѧʽΪNaxAlySiZOn��mH2O������Ϊ87��2g��{87��2g��(382��18m)g/mol}��1=0��2;���m=3����˸÷���ɸ�Ļ�ѧʽΪNaAlSi5O12��3H2O������ɸ�����ò�ͬ����ʽ��ʾ���ڱ仯�Ĺ����и�Ԫ�ص����ʵ����ıȲ��ܷ����仯�����������������ʽ��ʾΪNa2O��Al2O3��10SiO2��6H2O��
���㣺��������ķ��뷽�������ӷ���ʽ����д������ɸ�Ļ�ѧʽ��ȷ�������ӵļ�����֪ʶ��

 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�����ʵ�鷽������������
| A�������յķ�����������˿�Ͳ�˿ |
| B����ˮ���𱽺��Ҵ� |
| C��ʵ������N2��H2�Ʊ�NH3 |
| D���ñ���NaHCO3��Һ��ȥCO2�л��е�SO2���� |
�����16�֣�����ұ����ҵ�г��õ����Ӽ�����̼���̿���Ҫ�ɷ�ΪMnCO3�����������������ܵ�̼�������ʣ�Ϊԭ�����������̵Ĺ����������£�
��֪25�棬�������ʵ��ܶȻ��������£�
| ���� | Mn(OH)2 | Co(OH)2 | Ni(OH)2 | MnS | CoS | NiS |
| Ksp | 2.1��10��13 | 3.0��10��16 | 5.0��10��16 | 1.0��10��11 | 5.0��10��22 | 1.0��10��22 |
��2��������У�MnO2�����������¿ɽ�Fe2+��������ΪFe3+����Ӧ�����ӷ���ʽ�� ���Ӱ�ˮ������Һ��pHΪ5.0��6.0���Գ�ȥFe3+��
��3��������У�����2����Ҫ�ɷ��� ��
��4��������У��� ���������������������Mn���缫��Ӧ����ʽΪ ��
��5������ķ�ˮ�л�����Mn2+������ʯ�������һ�������õ�Mn(OH)2���������˺�������Һ�м�������Na2S�����ж���������Ϊ�˽�Mn2+����1.0��10��9mol/L����c(Na2S)������ mol/L��
Ϊ�������÷Ϸ�����������V2O5��VOSO4�������Բ�������������Ա����������һ�����ӽ��������շ����¹��գ���Ҫ�������£�
���ֺ���������ˮ�е��ܽ������£�
| ���� | VOSO4 | V2O5 | NH4VO3 | (VO2)2SO4 |
| �ܽ��� | ���� | ���� | ���� | ���� |
�ش��������⣺
��1����ҵ��V2O5ұ���������������ȼ������÷�Ӧ��������Ϊ_____________��
��2����Һ�к�������Ҫ�ɷ�Ϊ______________���ѧʽ����
��3���ù����з�Ӧ�۵ij����ʣ��ֳƳ����ʣ��ǻ��շ��Ĺؼ�֮һ���ò���Ӧ�����ӷ���ʽ____�������ʵĸߵͳ�����ҺpHӰ���⣬����Ҫ�����Ȼ��ϵ����NH4Cl������������Һ��V2O5�������ȣ����¶ȡ�������ͼ�ж���ѿ����Ȼ��ϵ�����¶�Ϊ___________��_________�档

��4���������ữ��H2C2O4��Һ�ζ�(VO2)2SO4��Һ���Բⶨ��Ӧ�ں���Һ�к���������Ӧ����ʽΪ��2VO2++H2C2O4+2H+��2VOn+ + 2CO2��+ mH2O������n��m�ֱ�Ϊ______________��________ ��
��5��ȫ��Һ����صĵ������ҺΪVOSO4��Һ����صĹ���ԭ��Ϊ��

��طŵ�ʱ�����ĵ缫��ӦʽΪ ��
��ҵ�����õ�����ࣨ��Ҫ����Fe2O3��CuO��Cr2O3�������������ʣ�����ͭ���Ƚ����������������£�
��֪�������ʳ�����pH��CaSO4���ܽ���������£�
| | Fe3+ | Cu2+ | Cr3+ |
| ��ʼ����pH | 2��1 | 4��7 | 4��3 |
| ��ȫ����pH | 3��2 | 6��7 | a |
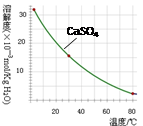
��1���ڽ��������г�������Fe2(SO4)3��Cr2(SO4)3��,��Ҫ����
��2���ڳ��������У���Ҫ��ȥFe3+��CaSO4,�������ز������ټ���ʯ�������pH�� ���ڽ���Һ���ȵ�80�棬 ��
��3��д����ԭ�����м���NaHSO3����Cu2O��������ӷ�Ӧ����ʽ ���˲����м���NaHSO3�õ�Cu2O�IJ���Ϊ95%����NaHSO3�����������˷��Լ��⣬������ֵ������� ��
��4��������Ũ�ȡ�1��10��5 mol?L-1��Ϊ������ȫ����ҪʹCr3+��ȫ������Ҫ����C(OH��)�� ����֪��Ksp[Cr(OH)3]=6��3��10-31��
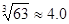 ��
�� 

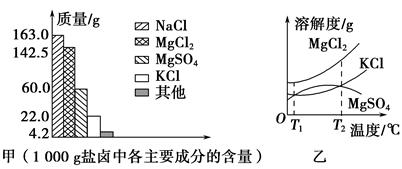


 10-12������ҺpH=6ʱ (��С���û�С�)Mg(OH)2����������
10-12������ҺpH=6ʱ (��С���û�С�)Mg(OH)2����������