��Ŀ����
12������һ������ˮ��Һ��ֻ���ܺ������������е������֣�K+��NH4+��Cl-��Ba2+��Fe3+��CO32-��SO42-����ÿ��ȡ100.00ml����ʵ�飮�ٵ�һ�ݼ���AgNO3��Һ�г���������
�ڵڶ��ݼ�����NaOH����ȣ��ռ�������0.896L����״̬�£���
�۵����ݼ�����BaCl2�ø������6.27g����������������ϴ�Ӹ����ʣ2.33g��
�Իش��й����⣺
��1��ͨ��ʵ��١��ڡ��ۺͱ�Ҫ���㣬��д�±��в������ӵ�Ũ�ȣ��ܼ�����ģ���д��������һ�������ڵ������0��������ȷ���Ƿ���ڵ������������
| �������� | Cl- | CO32- | SO42- | NH4+ | Ba2+ |
| c/��mol•L-1�� | ��ȷ�� | 0.2 | 0.1 | 0.4 | 0 |
���� �ټ���AgNO3��Һ�г���������˵����Һ�п��ܴ���Cl-��CO32-��SO42-��
�����ɵ�����Ϊ��������Һ��һ������NH4+���������ʵ���Ϊ$\frac{0.896L}{22.4L/mol}$=0.04mol��
��2.33g����Ϊ���ᱵ��6.27g����Ϊ���ᱵ��̼�ᱵ�Ļ����ٸ��ݵ���غ㣬�ó�һ�����ڼ����ӣ��ݴ˽��н��
��� �⣺�ٵ�һ�ݼ���AgNO3��Һ�г�����������AgNO3��Һ�г��������������У�Cl-��CO32-��SO42-��
�ڼ�����NaOH��Һ���Ȳ������壬�����ǰ��������ʵ���Ϊ��$\frac{0.896L}{22.4L/mol}$=0.04mol����Һ��һ������NH4+�������ʵ���Ϊ0.04mol��
�۲����������2.33gΪ���ᱵ�����ʵ���Ϊ��$\frac{2.33g}{233g/mol}$=0.01mol��6.27g���������ᱵ��̼�ᱵ��̼�ᱵ����Ϊ6.27g-2.33g=3.94g��̼�ᱵ�����ʵ���Ϊ��$\frac{3.94g}{197g/mol}$=0.02mol����ԭ��Һ��һ������CO32-��SO42-����һ��û��Ba2+��Fe3+��
��1������c=$\frac{n}{V}$����ó�c��CO32-��=$\frac{0.02mol}{0.1L}$=0.2mol/L��c��SO42-��=$\frac{0.01mol}{0.1L}$=0.01mol/L��c��NH4+��=$\frac{0.04mol}{0.1L}$=0.4mol/L�������ӵ�Ũ�Ȳ���ȷ����������Ũ��Ϊ0��
�ʴ�Ϊ��
| �������� | Cl- | CO32- | SO42- | NH4+ | Ba2+ |
| c/��mol•L-1�� | ��ȷ�� | 0.2 | 0.1 | 0.4 | 0 |
�ʴ�Ϊ���ǣ���СŨ��Ϊ0.2 mol•L-1��
���� ���⿼�鳣�����������ӵļ��鷽������Ŀ�Ѷ��еȣ�ע�����ճ������ӵĻ�ѧ���ʼ����鷽���������и��ݵ���غ��жϼ����ӵĴ���Ϊ�״��㣬����������ѧ�������Ӧ��������

 ���������������Բ��������ϵ�д�
���������������Բ��������ϵ�д���1��N2O5�뱽����������Ӧ���ɵ��������Ľṹ��ʽ��
 ��
����2��һ���¶��£��ں����ܱ�������N2O5�ɷ������з�Ӧ��2N2O5��g��?4NO2��g��+O2��g������H��0
�ٷ�Ӧ�ﵽƽ�������ͨ��һ������������N2O5��ת���ʽ����䣨���������С���������䡱����
�����Ϊ��Ӧ��T1�¶��µIJ���ʵ�����ݣ�
| t/s | 0 | 500 | 1000 |
| c��N2O5��/mol•L-1 | 5.00 | 3.52 | 2.48 |
����T2�¶��£���Ӧ1000sʱ���NO2��Ũ��Ϊ4.98mol•L-1����T2��T1���������=����
��3������H2��O2������Na2CO3��ɵ�ȼ�ϵ�ز��õ�ⷨ�Ʊ�N2O5��װ����ͼ��ʾ������YΪCO2��
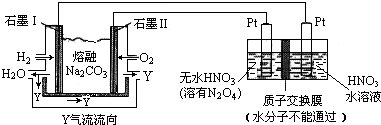
д��ʯī��缫�Ϸ�����Ӧ�ĵ缫��ӦʽH2+CO32--2e-�TH2O+CO2��N2O5�ڵ��ص����������ɣ��������������������
| A�� | �����£�1 mol ������������Ũ������ת�Ƶĵ�����Ϊ0 | |
| B�� | �ڱ�״���£�4.48 L�����ʺ��е���ԭ����ĿΪ0.4NA | |
| C�� | �ں���NA��CH3COO-�Ĵ�����Һ�У�H+��Ŀ�Դ���NA | |
| D�� | 0.1 mol N5+ ���к��еĵ�����Ϊ3.5NA |
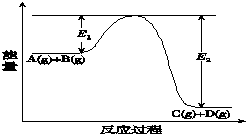 ��ӦA��g��+B��g��?C��g��+D��g�������е������仯��ͼ��ʾ��E1��0��E2��0�����ش��������⣮
��ӦA��g��+B��g��?C��g��+D��g�������е������仯��ͼ��ʾ��E1��0��E2��0�����ش��������⣮��ͼ��E1�����������Ƿ�Ӧ��Ļ�ܣ�
�÷�Ӧ�Ƿ��ȷ�Ӧ������ȡ����ȡ�������Ӧ�ȡ�H�ı���ʽΪE1-E2 ��
�ڵ���Ӧ�ﵽƽ��ʱ�������¶ȣ�A��ת���ʼ�С���������С�����䡱����
��2��800��ʱ����2L�ܱ������ڳ���0.50mol NO��0.25mol O2���������·�Ӧ��2NO��g��+O2��g���T2NO2��g����H��0����ϵ�У�n��NO����ʱ��ı仯���±���
| t/s | 0 | 1 | 2 | 3 | 4 | 5 |
| n��NO��/mol | 0.50 | 0.35 | 0.28 | 0.25 | 0.25 | 0.25 |
A��v��NO2����=v��O2������������B��������ѹǿ���ֲ���
C��v��NO����=2v��O2����D��������������ɫ����
����ʹ�÷�Ӧ�ķ�Ӧ����������ƽ��������Ӧ�����ƶ��Ĵ�ʩ��BC��
A���ʵ������¶� B����С��Ӧ���������C������O2��Ũ�ȡ�D��ѡ���Ч������
| A�� | ���ᣬ�������ͭ������ | B�� | ���ᣬ�ռ���ᱵ���ɱ� | ||
| C�� | ̼�ᣬ�Ҵ��������ƣ�ˮ | D�� | ���ᣬ��ʯ�ң��������أ������� |
| A�� | 3-����ȩ ��CH3��2CHCH2COH | B�� | TNT | ||
| C�� | Ӳ֬������� | D�� | ������� CH3OOCCH3 |