��Ŀ����
| |||||||||||||||||||||||||||
�𰸣�
������
������
(1) |
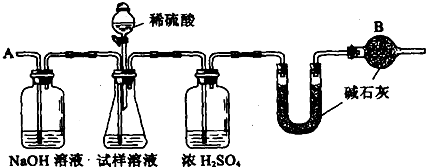
��Ʒ�أ������ᣬ������������ |
(2) |
��ֹ������CO2��ˮ��������U�� |
(3) |
ƫ�� |
(4) |
�ѷ�Ӧ������CO2ȫ������U���� |
(5) |
�жϷ�Ӧ������CO2�Ƿ�ȫ���ų�����U���ڼ�ʯ������ |
(6) |
|

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ