��Ŀ����
(8��)��һƿ������Һ�����ܺ���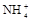 ��
��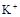 ��
�� ��
�� ��
�� ��
�� ��
��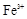 ��
�� ��
��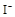 ��
��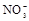 ��
��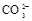 ��
��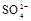 �е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺
�е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺
����pH��ֽ���飬������Һ��ǿ���ԣ�
��ȡ��������Һ���������� ������������ˮ������
������������ˮ������ ����Ϻ�ɫ��
����Ϻ�ɫ��
����ȡ������Һ����������μ���NaOH��Һ��ʹ��Һ�����Ա�Ϊ���ԣ��ڵμӹ�������
���ɰ�ɫ��������ȫ�ܽ⣻ȡ���ּ�����Һ���ȣ�������ų�����������ʹ��ʪ�ĺ�ɫʯ����ֽ������
����ȡ���֢��еļ�����Һ�������м��� ��Һ���а�ɫ�������ɡ�
��Һ���а�ɫ�������ɡ�
��������ʵ����ʵ�ش���������:
(1)����Һ�п϶����ڵ������� ���϶������ڵ������� ��
(2)����ۼ���NaOH��Һ�����������ɰ�ɫ��������ȫ�ܽ�����ӷ���ʽΪ ��
����8�֣���1��Ba2+��Al3+��I-��NH4+��Mg2+��Fe3+��CO23-��SO24-��NO-3 (ÿ��2�֣���4��) ��2��Al3+++3OH-=Al(OH)3�� Al(OH)3+OH-=[Al(OH)4]-��4�֣�
���������������ӹ��漰���ӷ���ʽ����д��
��1����Һ��ǿ���ԣ���һ��û��CO32���� ����Ϻ�ɫ��˵���е��ʵ����ɣ����һ�����е����ӣ����û��Fe3����NO3�����ڵμӹ����������ɰ�ɫ��������ȫ�ܽ⣬˵��һ�����������ӣ�һ��û��þ���ӣ�������ų�����������ʹ��ʪ�ĺ�ɫʯ����ֽ���������һ������NH4��������
����Ϻ�ɫ��˵���е��ʵ����ɣ����һ�����е����ӣ����û��Fe3����NO3�����ڵμӹ����������ɰ�ɫ��������ȫ�ܽ⣬˵��һ�����������ӣ�һ��û��þ���ӣ�������ų�����������ʹ��ʪ�ĺ�ɫʯ����ֽ���������һ������NH4��������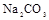 ��Һ���а�ɫ�������ɣ�����һ������Ba2������Ͳ��ܺ���SO42��������һ�����ڵ�������Ba2+��Al3+��I-��NH4+���϶������ڵ�������Mg2+��Fe3+��CO23-��SO24-��NO-3
��
��Һ���а�ɫ�������ɣ�����һ������Ba2������Ͳ��ܺ���SO42��������һ�����ڵ�������Ba2+��Al3+��I-��NH4+���϶������ڵ�������Mg2+��Fe3+��CO23-��SO24-��NO-3
��
(2)�������������������������Һǿ���У������йصķ���ʽ��
Al3+++3OH-=Al(OH)3�� �� Al(OH)3+OH-=[Al(OH)4]-��

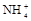 ��
��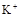 ��
��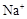 ��
�� ��
�� ��
�� ��
��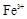 ��
�� ��
��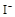 ��
��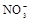 ��
��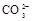 ��
��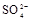 �е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺
�е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺 ������������ˮ������
������������ˮ������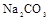 ��Һ���а�ɫ�������ɡ�
��Һ���а�ɫ�������ɡ� ��
��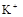 ��
��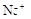 ��
��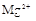 ��
��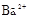 ��
��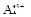 ����
����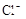 ��
��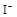 ��
��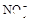 ��
�� ��
�� �е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺
�е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺 ������������ˮ������
������������ˮ������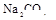 ��Һ���а�ɫ�������ɡ�
��Һ���а�ɫ�������ɡ� ��
��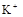 ��
�� ��
�� ��
��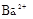 ��
��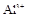 ����
����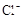 ��
�� ��
��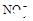 ��
��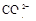 ��
��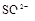 �е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺
�е�һ�ֻ��֡�ȡ����Һ��������ʵ�飺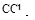 ������������ˮ������
������������ˮ������ ��Һ���а�ɫ�������ɡ�
��Һ���а�ɫ�������ɡ�