��Ŀ����
�����й���Һ������Ũ�ȵĹ�ϵʽ����ȷ����

A��c(Na��)��ͬ�Ģ�CH3COONa����NaHCO3����  ������Һ�е�pH���ۣ��ڣ���
������Һ�е�pH���ۣ��ڣ���
B��0.1mol��L-1ij��Ԫ����ǿ����NaHA��Һ�У�c(Na+)=2c(A2-)��c(HA-)��c(H2A)
C����ͼ��pH��7ʱ��c(Na+)��c(CH3COO-) ��c(OH-)��c(H+)
D����ͼ��a����Һ�и�����Ũ�ȵĹ�ϵ�ǣ�c(OH-)��c(H+)��c(CH3COO-)��2c(CH3COOH)
���𰸡�
AD
����������

��ϰ��ϵ�д�
�����Ŀ

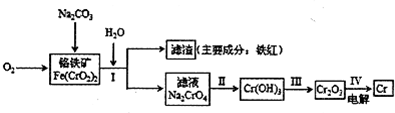
 Cr2O72-+H2O
Cr2O72-+H2O



