��Ŀ����
ij���᳧����β���е�SO2�Dz��ý�SO2ת��Ϊ��NH4��2SO4�ķ������ó�����������β���У�SO2���������Ϊ0.2%��O2���������Ϊ10%����β����8m3?h-1��������ͨ���Ƚ��������ȵ�400�棬��ͨ������ʹSO2ת��ΪSO3��Ȼ����һ�����ʵ�NH3��ϣ�����ˮʹ֮��Ӧ���ɣ�NH4��2SO4�����õ�����Һ�ڽᾧװ���н��д������ɵõ�����茶��塣�Իش���������⣺��1������Ӧ�е�����ֵSO2��O2�����ʵ���֮��Ϊ2��1���ó�������SO2��O2�����ʵ�����ʵ�ʱ�ֵΪ���٣�Ϊʲô��
��2������SO2��ȫת��ΪSO3��������ͬ������ͨ��NH3���������Ϊ���٣�
��3�����ó�ÿ���ŷ�10000m3����״�������ַ������������������㣬�ó�ÿ�£���30����㣩�ɵõ�����臨��ٶ֣�
��1����Ŀ��V��SO2����V��O2��=0.2%��10%=1��50��������������ԭ�����������۵�O2���������������SO2��ת���ʣ������ٴ��ŷŵ���Ⱦ��
��2��β����SO2������Ϊ8m3?h-1��0.2%=0.016m3?h-1�����ݹ�ϵʽ��2NH3��SO2����NH3���������Ϊ0.016m3?h-1 ��2=0.032m3?h-1��
��3��ÿ���ŷŵķ�����SO2�����Ϊ��
1��104m3��30��0.2%��103L?m-3=6��105L
���Ƶã�NH4��2SO4�������ɸ��������ϵʽ���㣺
2NH3��SO2����NH4��2SO4
��m�ۣ�NH4��2SO4��=
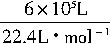 ��132g?mol-1��10-6t?g-1=3.536t��
��132g?mol-1��10-6t?g-1=3.536t��
��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ


