��Ŀ����
������H2����һ����̼��CO�������飨C8H18�������飨CH4�����Ȼ�ѧ����ʽ�ֱ�Ϊ��
H2��g��+![]() O2��g����H2O��l������H=��285.8kJ/mol
O2��g����H2O��l������H=��285.8kJ/mol
CO��g��+![]() O2��g����CO2��g������H=��283.0kJ/mol
O2��g����CO2��g������H=��283.0kJ/mol
C8H18��l��+![]() O2��g����8CO2��g��+9H2O��l������H=��5518kJ/mol
O2��g����8CO2��g��+9H2O��l������H=��5518kJ/mol
CH4��g��+2O2��g����CO2��g��+2H2O��l������H=��890.3kJ/mol
��ͬ������H2��CO��C8H18��CH4��ȫȼ��ʱ���ų��������ٵ��ǣ� ��
A��H2��g�� B��CO��g�� C��C8H18 ��l�� D��CH4��g��
B

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ

 ����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺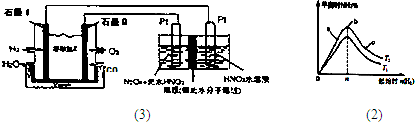
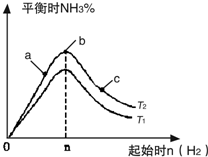 ����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺