��Ŀ����
9��ij�о�С��Ϊ��̽��һ�ֿ���ҩX���������ֶ�����Ԫ�أ�����ɺ����ʣ���Ʋ����������ʵ�飺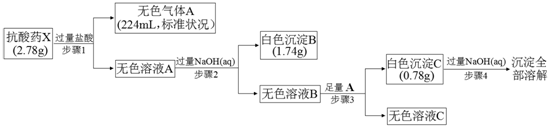
��ش��������⣺
��1����ɫ����B�н���Ԫ�ص����ӽṹʾ��ͼΪ
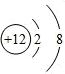 ��A�ĵ���ʽΪ
��A�ĵ���ʽΪ ��
����2������3�����ɳ���C�����ӷ���ʽΪAlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
��3��X�Ļ�ѧʽΪAlMg3��CO3����OH��7���ÿ���ҩ���ʺ�θ������֢����ʹ�ã�ԭ���Ǻ�θ�ᷴӦ���ɶ�����̼�������θ���ף�
��4�����ʵ�鷽����֤��ҺC�еĺ�̼����ȡ���������������Թ��У������Ȼ����������ڼ�������������ɫ��ζ�����壬ͨ�����ʯ��ˮ����ǣ�֤������̼��������ӣ�
���� ����ҩX�����������������ɫ����A����ɫ��ҺA����ҺA�м����������������Һ���˵õ���ɫ����B����ҺB����ҺB�м�������A�õ���ɫ����C������C�����������������Һ��ȫ�ܽ⣬˵��������CΪAl��OH��3������ҺB����ƫ�����ƣ�AΪCO2����ɫ��ҺCΪ̼��������Һ������ҩX�������ֶ�����Ԫ�أ��жϳ���Bֻ��ΪMg��OH��2����ɫ��ҺAΪAlCl3��Һ��MgCl2��Һ������ҩX�����������������ɫ���������̼��˵������CO32-��Al3+��Mg2+������n��Al3+��=n��Al��OH��3��=$\frac{0.78g}{78g/mol}$=0.01mol��n��Mg2+��=n��Mg��OH��2��=$\frac{1.74g}{58g/mol}$=0..03mol��n��CO32-��=n��CO2��=$\frac{0.224L}{22.4L/mol}$=0.01mol����õ�ʣ�ಿ������=2.78g-0.01mol��27g/mol-0.03mol��24g/mol-0.01mol��60g/mol=1.19g�����ݵ���غ㣬�����к���7������ɣ���һ��������=$\frac{1.19g}{7}$=0.17g���ж�ΪOH-���õ���ѧʽΪAlMg3��CO3����OH��7���ݴ˷����ش����⣮
��� �⣺����ҩX�����������������ɫ����A����ɫ��ҺA����ҺA�м����������������Һ���˵õ���ɫ����B����ҺB����ҺB�м�������A�õ���ɫ����C������C�����������������Һ��ȫ�ܽ⣬˵��������CΪAl��OH��3������ҺB����ƫ�����ƣ�AΪCO2����ɫ��ҺCΪ̼��������Һ������ҩX�������ֶ�����Ԫ�أ��жϳ���Bֻ��ΪMg��OH��2����ɫ��ҺAΪAlCl3��Һ��MgCl2��Һ������ҩX�����������������ɫ���������̼��˵������CO32-��Al3+��Mg2+������n��Al3+��=n��Al��OH��3��=$\frac{0.78g}{78g/mol}$=0.01mol��n��Mg2+��=n��Mg��OH��2��=$\frac{1.74g}{58g/mol}$=0..03mol��n��CO32-��=n��CO2��=$\frac{0.224L}{22.4L/mol}$=0.01mol����õ�ʣ�ಿ������=2.78g-0.01mol��27g/mol-0.03mol��24g/mol-0.01mol��60g/mol=1.19g�����ݵ���غ㣬�����к���7������ɣ���һ��������=$\frac{1.19g}{7}$=0.17g��n��OH-��=$\frac{1.19g}{17g/mol}$=0.07mol���ж�ΪOH-���õ���ѧʽΪAlMg3��CO3����OH��7��
��1������������֪��ɫ����B�н���Ԫ��ΪþԪ�أ�þ���Ӻ�����12�����ӣ��������������Ӳ㣬��һ��2�����ӣ��ڶ���8�����ӣ�þ���ӽṹʾ��ͼΪ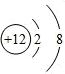 ��AΪ������̼���ڹ��ۻ����������̼Ϊֱ���ͽṹ�������д�������̼��˫����������̼�ĵ���ʽΪ
��AΪ������̼���ڹ��ۻ����������̼Ϊֱ���ͽṹ�������д�������̼��˫����������̼�ĵ���ʽΪ ��
��
�ʴ�Ϊ��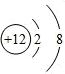 ��
�� ��
��
��2������3�����ɳ���CΪAl��OH��3����Ӧ�����ӷ���ʽΪAlO2-+CO2+2H2O=Al��OH��3��+HCO3-���ʴ�Ϊ��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
��3���������������֪XΪAlMg3��CO3����OH��7��X��θ�ᷴӦ���ɶ�����̼�������θ���ף�����Σ�գ�
�ʴ�Ϊ��AlMg3��CO3����OH��7����θ�ᷴӦ���ɶ�����̼�������θ���ף�
��4�����ʵ�鷽����֤��ҺC�еĺ�̼����ΪHCO3-��ȡ���������������Թ��У������Ȼ����������ڼ�������������ɫ��ζ�����壬ͨ�����ʯ��ˮ����ǣ�֤������̼��������ӣ�
�ʴ�Ϊ��ȡ���������������Թ��У������Ȼ����������ڼ�������������ɫ��ζ�����壬ͨ�����ʯ��ˮ����ǣ�֤������̼��������ӣ�
���� ���⿼��������ת���Ķ�����ϵ�����жϣ��������ʣ��������ʺͷ�Ӧ�����������仯�����ǽ���ؼ�����Ŀ�ѶȽϴ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д���֪����2R-COOH+2Na��2R-COONa+H2��
��R-COOH+NaHCO3��R-COONa+CO2��+H2O
| ʵ�鲽�� | ���ͻ�ʵ����� |
| ��1��ȡ9.0g�л���A����ʹ��������������ܶ�����ͬ������H2��45�� | ��1���ɴ˿�֪A����Է�������Ϊ90�� |
| ��2����9.0gA������O2�г��ȼ�գ���������λ���ͨ��Ũ���ᡢ��ʯ�ң����߷ֱ�����5.4g��13.2g�� | ��2���ɴ˿�֪A�ķ���ʽΪC3H6O3�� |
| ��3����ȡA9.0g����������NaHCO3��ĩ��Ӧ������CO2����2.24L��9.0gA�������������Ʒ�Ӧ����H2Ҳ��2.24L�����������Ϊ��״���²�ã��� | ��3���ɴ˿�֪A�к��й������У�����ṹ��ʽ��-COOH��-OH |
��4��A�ĺ˴Ź�����������ͼ�� | ��4���ɴ˿�֪A�к���4�ֲ�ͬ���ʵ���ԭ�� ��5�����ϣ�A�Ľṹ��ʽΪ  �� �� |
| A�� | 1��3-������ϩ�ȱ��ȶ� | |
| B�� | ����1��3-������ϩ�ȶ� | |
| C�� | 1��3-������ϩ���������ȷ�Ӧ | |
| D�� | ���������ɻ�����ķ�Ӧ��ֻ�����ʱ仯���������仯 |
| A�� | ������ΪCl2��Na | |
| B�� | ���������ܲ�������ɫ���� | |
| C�� | ���һ��ʱ���ȫ�����Һת�Ƶ��ձ��У�Ȼ��μӷ�̪��Һ����Һ�ʺ�ɫ | |
| D�� | ����ˮ�����������ռ����ϴ��������� |

| A�� | ����ͨ��������Ӧ�γɸ߷��ӻ����� | |
| B�� | ����ʹ����KMnO4��Һ��ɫ | |
| C�� | �ܷ����ӳɡ�ȡ������ȥ��������Ӧ | |
| D�� | 1 mol������������4 mol NaOH��Ӧ |
 ��
��
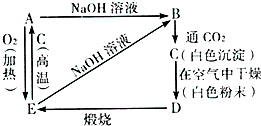 ������ͼ��Ӧ����仯���ش��й����⣮
������ͼ��Ӧ����仯���ش��й����⣮