��Ŀ����
��֪�������ݣ�
| �� �� | �۵�/�� | �е�/�� | �ܶ�/g��cm��3 |
| �� �� | ��114 | 78 | 0.789 |
| �� �� | 16.6 | 117.9 | 1.05 |
| �������� | ��83.6 | 77.5 | 0.900 |
| ŨH2SO4 |
| 338 | 1.84 |


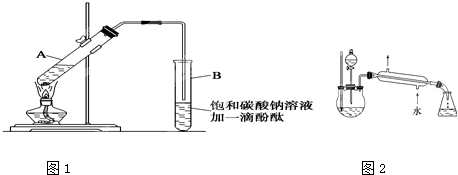
ʵ������������������Ҫװ������ͼI��ʾ����Ҫ����Ϊ������30mL�Ĵ��Թ��а������1��4��4�ı�������Ũ���ᡢ�Ҵ�������Ļ��Һ���ڰ���ͼI����װ�ã�ʹ����������������ͨ��ʢ15mL����Na2CO3��Һ(����1�η�̪��Һ)�Թ��Ϸ�2mm~3mm������С������Թ��еĻ��Һ���ܴ�С�Թ����ռ�Լ4mL����ʱֹͣ���ȣ�����С�Թܲ�������Ȼ���ã��ݷ��������������������
��ش��������⣺
��1��д��CH3COOH��CH3CH218OH��Ӧ�Ļ�ѧ����ʽ_____
ŨH2SO4��������__ _ ____ __
��2��������У���С������Թ��еĻ��Һ����ԭ��___ _____
��3����������۲쵽��������______ ___
��4��������У��������������ѡ�õ�������_________������Ӧ��_________�ڵ�����
��5��Ϊ������������IJ��ʣ��ס�����λͬѧ�ֱ����������ͼ�ס��ҵ�װ��(��ͬѧ����Ӧ�����ȴ�����ñ���Na2CO3��Һ��ȡ��ƿ�в���)������Ϊ����װ�ø�������Ϊʲô��
��
��1��CH3CH218OH+ CH3COOH����  CH3CO18OCH2CH3+ H2O
CH3CO18OCH2CH3+ H2O
��������ˮ��
��2�����ᡢ�Ҵ������������е�ӽ��ҽϵͣ������ȣ���Ӧ�����������ʧ
��3���ֲ㣬Na2CO3��Һ���ɫ��dz�������ݡ�
��4����Һ©�����ϣ�
��5���ң���Ӧ��������������

 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�2Fe��s��+O2��g��=2FeO��s����H=-544kJ?mol-1
4Al��s��+3O2��g��=2Al2O3��s����H=-3350kJ?mol-1
��2Al��s��+3FeO��s��=Al2O3��s��+3Fe��s���ġ�H�ǣ�������
| A��-859 kJ?mol-1 | B��+859 kJ?mol-1 | C��-1403 kJ?mol-1 | D��-2491 kJ?mol-1 |
 ij��ѧ��Ӧ�У��跴Ӧ���������ΪE1���������������ΪE2��
ij��ѧ��Ӧ�У��跴Ӧ���������ΪE1���������������ΪE2��